Answers
Answer:
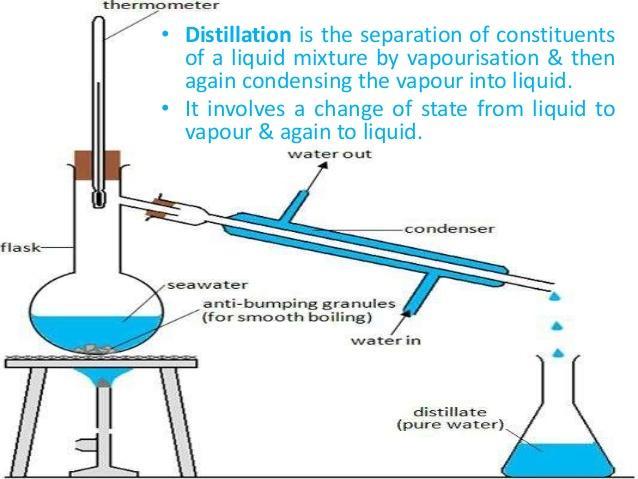
Distillation.
Explanation:
Hello there!
In this case, by considering the attached file, which depicts the process of distillation, whereby miscible liquids are separated out by firstly boiling and consequently condensing the liquid with the lowest boiling point so that the liquid with the highest boiling point remains in the flask. This process is widely used in the oil gas industry as lots of heavy (high boiling points) and light (low boiling points) produces can be obtained via distillation.
Regards!
Related Questions
Determine which base will work to deprotonate each compound in an acid/base extraction.
Benzene ring with a carboxylic acid on one carbon and a methyl ester group on the opposite carbon __________
Benzene ring with a three carbon chain attached to one carbon. At the other end of the three carbon chain is a hydroxyl group. ___________
Benzene ring with a hydroxyl group attached to one carbon and a tertbutyl group attached to the opposite carbon __________-
Answers
Answer:
A common method is to perform an acid-base reaction, which can ... acid is not particularly water-soluble due to its nonpolar aromatic ring, ... An acid-base extraction can be used to extract carboxylic acids from ... carbonic acid (H2CO3) can decompose to water and carbon dioxide gas.
Explanation:
The bases that will work to deprotonate the compounds are :
NaHCO₃ or NaOH.metal alkoxidesNaOHThe Benzene ring with carboxylic acid which is a weak acid can be deprotonated using NaOH or NaHCO₃ because of the weakness of the carboxylic acid.
Metal alkoxides like potassium tertiary butoxide can be used to deprotonate a Benzene ring with three carbon chain attached to a single carbon, and also containing a hydroxyl group is a very weak acid hence the use of Metal alkoxides.
Benzene ring with a hydroxyl group attached is also a weak acid just like the Benzene ring with carboxylic group hence it can also be deprotonated using NaOH
Learn more : https://brainly.com/question/24664427
How much natural gas does Canada have in reserve?
Answers
Answer:
Explanation:Marketable and technically recoverable resources
Canada's natural gas reserves are estimated to be 1,383 trillion cubic feet, of which 542 trillion cubic feet is conventional gas, and the rest is unconventional including coal-bed methane, shale and tight gas.
How many calories of energy are found in 8.3 grams or Fat? Every gram of fat
can produce 38 KJ of energy.
Answers
Answer: asd asd asd asdasdasdasd
CAN U JUST HELP ME PLS I NEED DESPERATE HELP. HELPPPP JUST PLS DO, IF U DON’T KNOW DON’T ANSWER :/
Answers
Answer:
i gotcha
Explanation:
-. How many moles of Pb are contained in 2.49 x 1025 atoms Pb?
Answers
Answer: There are [tex]4.13 \times 10^{-3}[/tex] moles of Pb are contained in [tex]2.49 \times 10^{25}[/tex] atoms Pb.
Explanation:
According to the mole concept, 1 mole of every substance contains [tex]6.023 \times 10^{23}[/tex] atoms.
Hence, number of moles present in [tex]2.49 \times 10^{25}[/tex] atoms of Pb are as follows.
[tex]No. of moles = \frac{2.49 \times 10^{25}}{6.023 \times 10^{23}}\\= 4.13 \times 10^{-3} mol[/tex]
Thus, we can conclude that there are [tex]4.13 \times 10^{-3}[/tex] moles of Pb are contained in [tex]2.49 \times 10^{25}[/tex] atoms Pb.
In redox reactions, the reactant that is oxidized is also called the _________.
a. oxidizing agent.
b. reducing agent.
c. reductant.
d. oxidant.
Answers
In redox reactions, the reactant that is oxidized is also called the reducing agent and is denoted as option B.
What is a Reducing agent?These are compounds which take part in a chemical reaction and lose their electrons in the process.
This leads to them becoming oxidized in a redox reaction and then acts as a reducing agent to the other compound.
Read more about Reducing agent here https://brainly.com/question/12549467
#SPJ2
there are two correct answers, reducing agent, and reductant
What is the molarity of a bleach solution
containing 9.5 g of Naoci per liter of
bleach
Answers
0.13M NaClO
Explanation:
9.5 g NaClO × (1 mol NaClO/74.442 g NaClO)
= 0.13 mol NaClO
Molarity = 0.13 mol NaClO/1 L
= 0.13M NaClO
True or False: Both molecules and compounds are pure substances.
Answers
Answer:
False
Explanation:
Hope this helps!
Answer:
True
Explanation:
It's because both compounds and elements are considered pure substances and a molecules is considered an element.
The Atlantic ocean grows in size due to a
A. thrust boundary
(B. divergent boundary)
C. transform boundary
D. convergent boundary
Answers
Answer:
B. Divergent boundary
Hope this helps :)
why is there need to standardize a solution
Answers
standardization of a volumetric solution used for titration is one of the most important preconditions for reliable and transparent titration results.
Calculate the number of joules of heat energy needed to increase the temperature of 25.0 g of metal from 21.0 ºC to 80.0 ºC. The specific heat of the metal is 0.521 J/gºC.
Answers
Answer:
Q = 768.47 J
Explanation:
Given that,
Mass of the metal, m = 25 g
Initial temperature, T₁ = 21.0 ºC
Final temperature, T₂ = 80.0 ºC
The specific heat of the metal is 0.521 J/gºC.
We know that the heat released due to the change in temperature is given by :
[tex]Q=mc\Delta T\\\\=25\times 0.521\times (80-21)\\Q=768.47\ J[/tex]
Hence, 768.47 J of heat energy will be needed.
What compound of
calcium is used
for makings refraction furnace linings
Answers
A helium balloon has a volume of 25 L when the pressure is 1.551 ATM and the temperature is 20°C. The balloon is cooled at a constant pressure until the temperature is -11°C. What is the volume of the balloon at this stage?
Answers
Answer:
The balloon is cooled at a constant pressure until the temperature is -11°C. What is the volume of the balloon at this stage?
Which chemical reaction is the slowest?
A. Cake rises as it cooks in the oven.
B. Drain cleaner fizzes up as it encounters blockages in a pipe.
C. Baking soda and vinegar react.
D. Milk turns sour.
WILL GIVE BRAINLIEST!!
Answers
Answer:
Milk turns sour
The chemical reaction or chemical change which is slowest is milk turning sour as it involves enzymatic actions which are slow reactions.
What is a chemical change?Chemical changes are defined as changes which occur when a substance combines with another substance to form a new substance.Alternatively, when a substance breaks down or decomposes to give new substances it is also considered to be a chemical change.
There are several characteristics of chemical changes like change in color, change in state , change in odor and change in composition . During chemical change there is also formation of precipitate an insoluble mass of substance or even evolution of gases.
There are three types of chemical changes:
1) inorganic changes
2)organic changes
3) biochemical changes
During chemical changes atoms are rearranged and changes are accompanied by an energy change as new substances are formed.
Learn more about chemical changes,here:
https://brainly.com/question/23693316
#SPJ5
on the planet mars the average night temperature is 24k. what is the temperature in degrees celsius and degree fahrehheit
Answers
What is the MOST LIKELY reason that they build these layers? A. Help them capture food because their calcium carbonate exterior keeps them from drying out in their specific environment. B. Help them capture food because their calcium carbonate exterior allows them to withstand an attack from their predators in their specific environment. C. Protect them from predators because their calcium carbonate exterior allows them to withstand an attack from their predators in their specific environment. D. Protect them from predators because their calcium carbonate exterior keeps them from drying out in their specific environment.
Answers
Answer:
C. Protect them from predators because their calcium carbonate exterior allows them to withstand an attack from their predators in their specific environment.
Explanation:
They build layers to protect themselves from predators. Plus, I've seen this question and got it right.
How much heat energy is required to boil 66.7 g of ammonia, NH3? The molar heat of vaporization of ammonia is 23.4 kJ/mol.
Answers
Answer:
91.7 kJ
Explanation:
Step 1: Given data
Mass of ammonia (m): 66.7 gMolar heat of vaporization of ammonia (ΔH°vap): 23.4 kJ/molStep 2: Calculate the moles (n) corresponding to 66.7 g of ammonia
The molar mass of ammonia is 17.03 g/mol.
66.7 g × 1 mol/17.03 g = 3.92 mol
Step 3: Calculate the heat (Q) required to boil 3.92 moles of ammonia
We will use the following expression.
Q = ΔH°vap × n
Q = 23.4 kJ/mol × 3.92 mol = 91.7 kJ
A mole of a substance has a mass in grams that is equal to the molecular mass. For example, a carbon atom has a mass of 12.01 u. A mole of carbon has a mass of 12.01 g. Based on their molecular masses, list the molar massof each substance:Fe2O3__________CO __________Fe __________CO2__________ slader
Answers
Answer:
M(Fe₂O₃) = 159.70 g/mol
M(CO) = 28.01 g/mol
M(Fe) = 55.85 g/mol
M(CO₂) = 44.01 g/mol
Explanation:
We can calculate the molar mass of a compound by summing the molar masses of the elements that form it.
Fe₂O₃
M(Fe₂O₃) = 2 × M(Fe) + 3 × M(O) = 2 × 55.85 g/mol + 3 × 16.00 g/mol = 159.70 g/mol
CO
M(CO) = 1 × M(C) + 1 × M(O) = 1 × 12.01 g/mol + 1 × 16.00 g/mol = 28.01 g/mol
Fe
M(Fe) = 1 × M(Fe) = 1 × 55.85 g/mol = 55.85 g/mol
CO₂
M(CO₂) = 1 × M(C) + 2 × M(O) = 1 × 12.01 g/mol + 2 × 16.00 g/mol = 44.01 g/mol
Observe the above circuit diagram, if the gap is filled with a plastic wire, will the bulb glow? Justify your answer
Answers
Answer:
YES THE BULB WILL GLOW.
Explanation:
BECAUSE THE CIRCUIT UPPER AREA IS MADE OF PLASTIC SO THE PLASTIC CONDUCTS THE ELECTRICITY SO THE BULB WILL GLOW.
The combustion of 3.795 mg of liquid B, which contains only C, H, and O, with excess oxygen gave 9.708 mg of CO2 and 3.969 mg of H2O. In a molar mass determination, 0.205 g of B vaporized at 1.00atm and 200.0'C and occupied a volume of 89.8 mL. Derive the empirical formula, molar mass, and molecular formula of B and draw three plausible structures
Answers
Answer:
Explanation:
9.708 mg of CO₂ will contain 12 x 9.708 / 44 = 2.64 g of C .
3.969 mg of H₂O will contain 2 x 3.969 / 18 = .441 g of H .
mg of O in given liquid B = 3.795 - ( 2.64 + .441 ) = .714 mg
ratio of mg of C , H , O in the compound = 2.64 : .441 : .714
ratio of no of atoms of C , H , O in the compound
= 2.64 / 12 : .441 /1 : .714 / 16
= .22 : .44 : .0446
= .22 / .22 : .44 / .22 : .044 / .22
= 1 : 2 : .2
1 x 5 : 2 x 5 : .2 x 5
= 5 : 10 : 1
empirical formula of the compound = C₅H₁₀O
Volume of 89.8 mL at 1 .00 atm at 200⁰C
volume of gas at 1 atm and 0⁰C = 89.8 x 273 / 473 mL
= 51.83 mL
51.83 mL weighs .205 g
22400 mL will weigh .205 x 22400 / 51.83 g
= 88.6 g
So molecular weight = 88.6
Let molecular formula be (C₅H₁₀O)ₙ
molecular weight = n ( 5 x 12 + 10 + 16 )
= 86 n
86 n = 88.6
n = 1 approx
So molecular formula is same as empirical formula
C₅H₁₀O is molecular formula .
How many liters of C3H6O are present in a sample weighing 25.6 grams?
Answers
To Find :
Number of moles of C₃H₆O present in a sample weighing 25.6 grams.
Solution :
Molecular mass of C₃H₆O is :
M = (6×12) + (6×1) + (16×1) grams
M = 94 grams/mol
We know, number of moles of 25.6 grams of C₃H₆O is :
[tex]n = \dfrac{Given \ Mass \ Of \ C_3H_6O }{Molar\ Mass \ Of \ C_3H_6O }\\\\n = \dfrac{25.6}{94}\ mole\\\\n = 0.27 \ mole[/tex]
Hence, this is the required solution.
How does high pressure effect the solubility of a gas?
Answers
Answer:
Gases as might be expected, increase in solubility with an increase in pressure. Henry's Law states that: The solubility of a gas in a liquid is directly proportional to the pressure of that gas above the surface of the solution. ... When the bottle is opened, the pressure above the solution decreases.
Answer:High pressure increases the solubility.
Explanation: i just took the quiz
If you wanted to make .5 L of a 1 mole/L (M) of MgSO4 solution, how many grams of MgSO4 would you use?
Answers
Answer:
0.5 grams
Explanation:
What eventually happens to gas if its pressureis increased?
Answers
Answer:
the rate increases
Explanation:
they are closer to each other, they will collide with each other more frequently and more successful collision
The coordination compound Co3[Cr(CN)6]2 contains Co2+ cations and a complex anion. What is the likely oxidation state for Cr in the anion?
Answers
Answer:
-3
Explanation:
The oxidation state or oxidation number of an atom is the total number of electrons that an atom either gains or loses in order to form a chemical bond with another atom.
The complex anion here is [Cr(CN)6]3-.
Now, as the oxidation state of CN or cyanide ligand is -1, and if we suppose the oxidation state of Cr to be 'x', then; x - 6 = -3 (overall charge on the anion),
so x= +3. Hence the oxidation state of Chromium in this complex hexacyanochromium (III) anion comes out to be -3.
.
1. How many moles of calcium are in 525g Ca?
Answers
Answer:
13.1743
Explanation:
1 grams Calcium to mol = 0.02495 mol
According to the mole concept, there are 13.12 moles of calcium in 525 g of calcium.
What is a mole?Mole is defined as the unit of amount of substance . It is the quantity measure of amount of substance of how many elementary particles are present in a given substance.
It is defined as exactly 6.022×10²³ elementary entities. The elementary entity can be a molecule, atom ion depending on the type of substance. Amount of elementary entities in a mole is called as Avogadro's number.
It is widely used in chemistry as a suitable way for expressing amounts of reactants and products.For the practical purposes, mass of one mole of compound in grams is approximately equal to mass of one molecule of compound measured in Daltons. Molar mass has units of gram per mole . In case of molecules, where molar mass in grams present in one mole of atoms is its atomic mass.Number of moles= mass/molar mass= 525/40=13.12 moles.
Thus, there are 13.12 moles of calcium in 525 g of calcium.
Learn more about mole,here:
https://brainly.com/question/26416088
#SPJ2
A substance with a lower specific heat capacity would:
1. Heat up faster and cool down slower
2. Heat up faster and cool down faster
3. Heat up slower and cool down faster
4. Heat up slower and cool down slower
Answers
Answer:
i think 2
Explanation:
i only think 2 i only think
Select the keyword or phrase that will best complete each sentence. A Claisen reaction is a nucleophilic ____________ in which an enolate is the nucleophile.
Answers
Answer:
Substitution.
Explanation:
Claisen reaction was first published in 1887 by a prominent German chemist known as Rainer Ludwig Claisen.
A Claisen reaction is a nucleophilic substitution in which an enolate is the nucleophile. It's typically a reaction of two molecules of an ester to form a β-keto ester, in the presence of an alkoxide base. Thus, a Claisen reaction is simply a characteristic condensation reaction of esters through a nucleophilic carbonyl substitution with an enolate such as a ketone enolate or an ester enolate.
Furthermore, a Claisen reaction results in the formation of a carbon-carbon bond in the presence of a strong base to yield a β-keto ester.
How many grams of ethanol, C2H5OH, can be boiled with 843.2 kJ of heat energy? The molar heat of vaporization of ethanol is 38.6 kJ/mol.
Answers
Answer:
1.00 × 10³ g
Explanation:
Step 1: Given data
Provided heat (Q): 843.2 kJMolar heat of vaporization of ethanol (ΔH°vap): 38.6 kJ/molStep 2: Calculate the moles of ethanol vaporized
Vaporization is the passage of a substance from liquid to gas. We can calculate the number of moles (n) vaporized using the following expression.
Q = ΔH°vap × n
n = Q / ΔH°vap
n = 843.2 kJ / (38.6 kJ/mol) = 21.8 mol
Step 3: Calculate the mass corresponding to 21.8 moles of ethanol
The molar mass of ethanol is 46.07 g/mol.
21.8 mol × 46.07 g/mol = 1.00 × 10³ g
The conversion of the liquid into water vapor is called vaporization.
The correct answer to the question is [tex]1.00 * 10^3 g[/tex]
The data given in the question is as follows:-
Provided heat (Q): 843.2 kJ Molar heat of vaporization of ethanol (ΔH°vap): 38.6 kJ/molThe formula we will use to calculate the moles is as:-
[tex]Q = H^ovap* n\\n = \frac{Q}{H^ovap}\\n = \frac{843.2 kJ}{(38.6 kJ/mol)} \\\\= 21.8 mol[/tex]
To calculate the mass of the following is as follows:-
The molar mass of ethanol is 46.07 g/mol.
[tex]21.8 mol* 46.07 g/mol = 1.00 * 10^3 g[/tex]
Hence the correct answer is [tex]1.00 * 10^3 g[/tex].
For more information, refer to the link:-
https://brainly.com/question/22810476
Atmospheric pressure decreases as altitude increases. In other words, there is more air pushing down on you at sea level, and there is less air pressure pushing down on you when you are on a mountain.If pentane (C5H12), hexane (C6H14), and hexanol (C6H13OH) are heated evenly at different altitudes.
Required:
Rank them according to the order in which you would expect them to begin boiling.
Answers
Answer:
Pentane » Hexane » Hexanol
Explanation:
Pentane and Hexane are simple molecular structured with discrete molecules having weak vanderwaals forces of attraction. But Hexane has high molecular mass than pentane hence pentane has lower boiling point than Hexane.
Hexanol has hydrogen bonds between highly electronegative oxygen atom and the hydrogen atom which break on high heat energy application hence it has high boiling point.