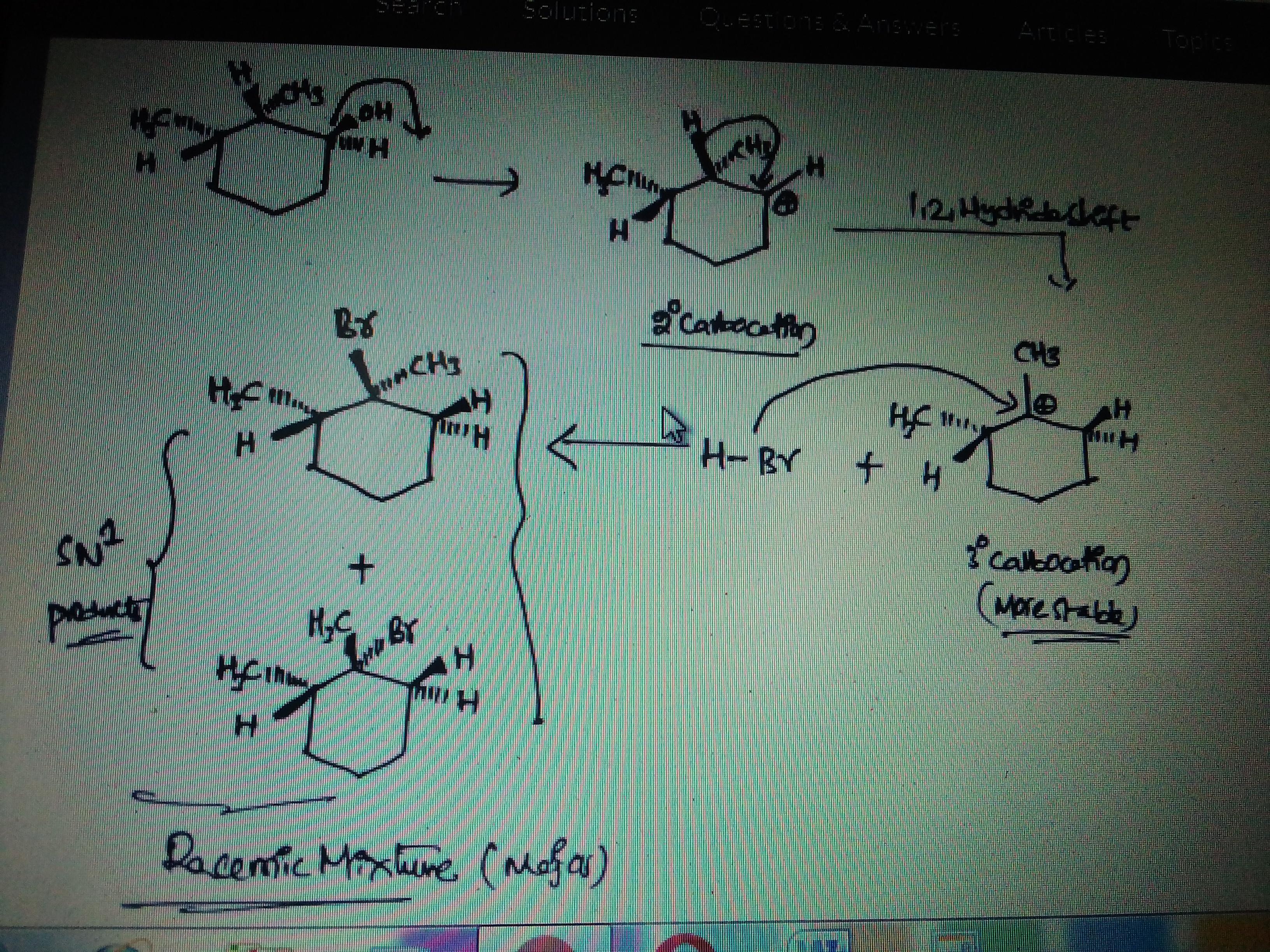
This molecule undergoes a substitution reaction through an SN1 pathway when warmed and stirred with HBr. Draw the two substitution products of this reaction. Show the correct stereochemistry by using wedges and dashes at all chiral centers. In part two, select the term that correctly describes the relationship between the products.
Answers
Answer:
See explanation and image attached
Explanation:
The mechanism of this reaction is shown in the image attached. The image was obtained from HomeworkLib.
The first step in the reaction is protonation and loss of -OH2. A carbocation is now created which undergoes a 1,2 hydride shift. This yields a tertiary carbocation which is more stable.
A racemic mixture of two products, major and minor are now obtained as shown.
Related Questions
What are similarities and differences between a falling object and a pendulum?
Answers
Answer:
The similarity is that they both follow the laws of physics.
In both cases, the motion is opposed by the medium where the object presses through the medium and forces it to part and flow around the object. This creates friction.
If an object is smooth and pointed like a bomb, the friction is minimised. If it is rough with pockets or vertical flat surfaces, the friction is maximised. A parachute presents a round hollow cup face to the flow, maximising friction. This is due to a hole in the top of the parachute.
The difference is the power source. In the vertical direction it is generated by gravity, the force created by the planet. This force is opposed not only by friction but also the difference between the density of the object and the medium surrounding it. If there is a large difference then the acceleration will be large and a high percentage of g.
For example we have a density of 1000 kg/meters cubed and air has a density of 1 Kg per meters cubed, the difference is 999, so we fall at an acceleration of 999/1000 x g. minus the effects of friction.
In the horizontal direction, gravity still opposes the movement creating friction in the vertical direction such as dragging a heavy object on the floor behind you. Force needs to be created horizontally to oppose the friction and to create an acceleration. A balance is soon reached where the acceleration stops and the object is dragged at a constant velocity.
Consider the Fischer ester synthesis of methyl benzoate from benzoic acid and methanol in the presence of sulfuric acid as a catalyst. A reaction was performed in which 3.8 g of benzoic acid was reacted with excess methanol to make 2.0 g of methyl benzoate. Calculate the theoretical yield and percent yield for this reaction.
Answers
Answer:
48.8%
Explanation:
The reaction has a 1:1 mole ratio so;
Number of moles of benzoic acid reacted = mass/molar mass = 3.8 g/122.12 g/mol = 0.03 moles
So;
0.03 moles of methyl benzoate is formed in the reaction
Mass of methyl benzoate formed = 0.03 moles * 136.15 g/mol = 4.1 g
percent yield = actual yield/theoretical yield * 100/1
percent yield = 2.0 g/4.1 g * 100 = 48.8%
150. g of aluminum chloride in 0.450 liters of solution, what is the concentration? (any examples are helpful, thank you)
Answers
Answer:
2.49 M
Explanation:
Step 1: Given data
Mass of AlCl₃: 150. gVolume of solution: 0.450 LStep 2: Calculate the moles AlCl₃ (solute)
The molar mass of AlCl₃ is 133.34 g/mol.
150. g × 1 mol/133.34 g = 1.12 mol
Step 3: Calculate the molar concentration of AlCl₃
Molarity is equal to the moles of solute divided by the liters of solution.
M = 1.12 mol/0.450 L = 2.49 M
Which of the following safety devices ensures that electricity will flow through it when flowing "downhill" instead of through people? A. Surge protectors B. Electric generators C. Ground wires D. Special plugs for high-current devices
Answers
Answer:
Ground Wires
Explanation:
14. Explain, in terms of ions, why a 10.0-milliliter sample of 0.10 M NH4Cl(aq) is a poorer conductor of
electricity than a 10.0-milliliter sample of the 0.30 M NH4Cl(aq).
Neeed now
Answers
One of the methods that scientists can use to determine the toxicity of a chemical compound is to perform a dose-response analysis.
a. True
b. False
Answers
Answer:
a. True
Explanation:
Hello there!
In this case, since the dose-response method is crucial concept in toxicology as it exhibits the result of exposures to toxic chemical compounds with changes in body functions or health, this statement is a. True , as this analysis is understood by means of the relationship -the higher the dose, the more severe the response-, and of course, it is based on observed data from controlled experiments in animal, human clinical, or cell studies, in the light of the determination of the toxicity of the analyzed chemical.
Regards!
Which simple machine is best used to split apart an object
A. A screw
B. A lever
C. A wedge
D. A pulley
Answers
Answer:
its A screw :))) your welcome
How can we find life on other planets?
Answers
Through satellites...
A student was directed to convert the mass of a boulder from kg to pounds.
Conversion factor: 1 pound = 454 g
The mass is 22 kg. The student calculated the weight to be 0.048 pounds.
Is this correct? If not, what went wrong?
Answers
Answer:
Incorrect
Explanation:
Given that,
The mass of a boulder, m = 22 kg
We need to find the weight of the student in pounds.
We know that,
1 pound = 454 g
or
1 g = (1/454 pounds)
22 kg = 0.022 grams
So,
0.022 grams = [tex]\dfrac{0.022}{454}\ pounds[/tex]
= 0.000048 pounds
The student says the weight is 0.048 pounds. It means he was incorrect.
Which type of light would make an object appear larger and closer?
Answers
Answer:
A convex lens bends light rays inward, which results in the object being perceived as larger or closer.
Explanation:
The light ray refracted from an object may look different based on how they bends light and at what angle. Convex lens bends light inwards and the refracted light appear closer and bigger whereas, a concave lens makes light ray away and smaller.
What is refraction?Refraction is the optical phenomenon by which a light ray bends in its direction when passing from one medium to the other. For instance light ray bends when it pass from air to water or air to glass and vice versa.
The measure of bending of light in a medium is called refractive index of that medium. Refraction makes the object appear to be closer sometimes such as in water when light bends in direction.
When light refracts from convex lens, the bending is inwards and the object is perceived to be closer and larger. Whereas, a concave lens make the bending outwards where the object appeared to be smaller and far.
To find more on refraction, refer here:
https://brainly.com/question/14760207
#SPJ2
True or false, rewrite it to make it true.
To balance a chemical equation, only the subscripts of a reactant and product can be changes
Answers
Answer:
false
Explanation:
Is wrought iron substitutional or interstitial?
Answers
Answer:
interstitial is correct my child
Explanation:
The distance that is covered by the wave in one second is called
Answers
Answer:
The distance that is covered by the wave in one second is called frequency.
Explanation:
How many grams of oxygen are needed to burn 10.0 moles of C3H8?
Answers
which type of neuron carries impulses from the brain?
Answers
Answer:
Using my searching skills, the answer is motor neurons.
Explanation:
Motor neurons carry impulses from the brain and spinal cord to muscles or glands.
Joseph rides his bike for 30 miles in one hour
going west . Is it speed velocity or acceleration
Answers
Answer:
velocity
Explanation:
because he is going 30 miles west
CH4 Structural formulas
Answers
Explanation:
Methane = CH4(formula)
need this ASAP
Which of these must be FALSE, in order for evolution by natural selection to occur?
A. Variation between individuals exists in a population.
B. Particular variations of traits are passed on from one generation to the next.
C. Resources are limited, leading to competition within the population.
D. The particular traits present in one generation have no effect on traits in the next generation.
thank you :D
Answers
igneous sedimentary, and metamorphic rocks are found at the same cliff side.Which statement is correct?
Answers
Answer:
The metamorphic rock and sedimentary rock were already at this location and magma flowed into other rocks
Explanation:
How would you prepare 500 cm3 of 2.0 M solution of Na2CO3 solution from its anhydrous salt? [C = 12, O = 16, Na = 23
Answers
Answer:
The answer to your question is given below
Explanation:
We'll begin by converting 500 cm³ to L. This can be obtained as follow:
1000 cm³ = 1 L
Therefore,
500 cm³ = 500 cm³× 1 L / 1000 cm³
500 cm³ = 0.5 L
Next, we shall determine the number of mole of Na₂CO₃ in the solution. This can be obtained as follow:
Volume = 0.5 L
Molarity = 2 M
Mole of Na₂CO₃ =?
Molarity = mole /Volume
2 = Mole of Na₂CO₃ / 0.5
Cross multiply
Mole of Na₂CO₃ = 2 × 0.5
Mole of Na₂CO₃ = 1 mole
Finally, we shall determine the mass of 1 mole of Na₂CO₃. This can be obtained as follow:
Mole of Na₂CO₃ = 1 mole
Molar mass of Na₂CO₃ = (23×2) + 12 + (16×3)
= 46 + 12 + 48
= 106 g/mol
Mass of Na₂CO₃ =?
Mass = mole × molar mass
Mass of Na₂CO₃ = 1 × 106
Mass of Na₂CO₃ = 106 g
Thus, to prepare the solution, weigh 106 g of Na₂CO₃ and dissolve in 500 cm³ (i.e 0.5 L) of water.
Please help!
Karl-Anthony is trying to plate gold onto his silver ring. He constructs an electrolyte cell using his ring as one of the electrodes. He runs this cell for 90.6 minutes at 213.8 mA. How many moles of electrons were transferred in this process?
Answers
Answer:
0.012 mole of electron
Explanation:
From the question given above, the following data were obtained:
Time (t) = 90.6 minutes
Current (I) = 213.8 mA
Number of mole of electrons =?
Next, we shall convert 90.6 mins to seconds. This can be obtained as follow:
1 min = 60 s
Therefore,
90.6 mins = 90.6 × 60
90.6 mins = 5436 s
Next, we shall convert 213.8 mA to A. This can be obtained as follow:
1000 mA = 1 A
Therefore,
213.8 mA = 213.8 mA × 1 A / 1000 mA
213.8 mA = 0.2138 A
Next, we shall determine the quantity of electricity used in the process. This can be obtained as follow:
Time (t) = 5436 s
Current (I) = 0.2138 A
Quantity of electricity (Q) =?
Q = it
Q = 0.2138 × 5436
Q = 1162.2168 C
Next, the equation for the reaction.
Au⁺ + e —> Au
From the balanced equation above,
1 mole of electron was transferred.
Recall:
1 faraday = 96500 C = 1 e
Thus,
96500 C of electricity is needed to transfer 1 mole of electron.
Finally, we shall determine the number of mole electrons transferred by the application of 1162.2168 C of electricity. This can be obtained as follow:
96500 C of electricity is needed to transfer 1 mole of electron.
Therefore,
1162.2168 C of electricity will transfer = 1162.2168 / 96500 = 0.012 mole of electron
Thus, 0.012 mole of electron was transferred in the process.
Following are the calculation to the moles of electrons that transferred in the process:
Given
Current flows
time[tex]= 90.6\ min = 90.6 \times 60\ sec= 5436 \ sec\\\\[/tex]
For Step 1:
Calculating the Total charge flown in the given time:
For Step 2:
Charge on a single electron
So:
Following are the calculation to the charges of electrons:
Following are the calculation to the electrons transferred:
[tex]=\frac{(726.38\times 10^{19})}{ (6.023 \times 10^{23})}\\\\= 120.60\times 10^{-4}\ moles\\\\[/tex]
Learn more:
brainly.com/question/12161663
Both the sweatshirt and the balloon are negatively charged. They are most
likely to (blank)
one another.
A-give
B-attract
C-repel
D-help
Answers
Answer:
B attract
Explanation:
There will be statication
Help plzz pick on the picture
Answers
Which factor has the greatest effect on object X remaining in its orbit?
A:The size of Earth compared to object X
B:The gravitational force between the Earth and object X
C:The gravitational force between the Sun and object X
D: The magnetic field between the Sun and object X.
Answers
Explanation:
Stop cheating and use your brains kiddos
how many moles are 20 grams of Helium ?
Answers
Answer: The correct answer is 5.00 moles
Explanation:
Aqueous hydrochloric acid HCl will react with solid sodium hydroxide NaOH to produce aqueous sodium chloride NaCl and liquid water H2O. Suppose 15.7 g of hydrochloric acid is mixed with 27. g of sodium hydroxide. Calculate the maximum mass of sodium chloride that could be produced by the chemical reaction.
Answers
Answer:
25.155 gram
Explanation:
HCl(aq.) + NaOH(aq.) ⇒ NaCl + H2O
According the above equation equal number of moles of reactant are producing same number of moles of products.
Now, no. mole of HCl = 15.7/(1+35.5) = 0.43 moles
no. of moles of NaOH = 27/(23+16+1) = 0.675 moles
Since the amount of sodium hydroxide exceeds the amount of hydrochloric acid, the maximum amount of each substance participating in the reaction is HClν ν 0.43 moles .Therefore, the maximum amount of NaCl that can be produced is as follow
= (23+35.5)×0.43 = 25.155 gram
Match each object to its definition.
A. meteorite
B. meteoroid
C. meteor
->
1. The remains of a rock from space that strikes Earth’s surface
2. A rock that burns up in Earth’s atmosphere
3. A small piece of rock in space
Answers
Answer:
definition 1. goes with B. meteoroid
definition 2. goes with C. meteor
definition 3. goes with A. meteorite
Answer: meteoroid - a small piece of rock in space, meteor - a rock that burns up in Earths atmosphere, meteorite - The remains of a rock from space that strike Earth's surface.
Explanation:
It’s worth 50 points! Please help!
Answers
Answer:
1) NO2
2) 49.4%
Explanation:
water
air
multiple cells
a place to live
Answers
plants need to be watered in order to survive
Answer:
I guess it WATER
does anyone know the answer to this