Answers
Answer:
Explanation:
Molarity of NaOAc needed
Using the Henderson-Hasselbalch Equation calculate base molarity needed given [HOAc] = 1.00M and pKa(NaOAc) = 4.75 and [HOAc] = 1.00m.
pH = pKa + log [NaOAc]/[HOAc]
5.00 = 4.75 + log[NaOAc]/[1.00M]
5.00 - 4.75 = log [NaOAc] - log[1.00M]
log [NaOAc] = 0.25 => [NaOAc] = 10⁰·²⁵ M = 1.78
Given 10ml of HOAc, how much (ml) 1.78M NaOAc to obtain a buffer pH of 5.00.
Determine Volume of Base Needed
(M·V)acid = (M·V)base => V(base) = (M·V)acid / (M)base
Vol (NaOAc) needed = (1.00M)(0.010L)/(1.78M) = 0.0056 liter = 5.6 ml.
Checking Results:
5.00 = 4.75 + log [1.78M]/[1.00M] = 4.75 + 0.25 = 5.00 QED.
The volume of 1.00 M sodium acetate solution needed to prepare an acetic/acetate buffer of pH 5.00 using 10.0 mL of 1.00M acetic acid solution is 17.8 mL.
We can find the volume of the acetate solution with the Henderson-Hasselbalch equation:
[tex]pH = pka + log(\frac{[CH_{3}COONa]}{[CH_{3}COOH]})[/tex] (1)
Where:
[CH₃COOH] = 1.00 M
[CH₃COONa] =?
pH = 5.00
pKa = 4.75
From equation (1), we have:
[tex] log(\frac{[CH_{3}COONa]}{[CH_{3}COOH]}) = pH - pKa [/tex]
[tex] \frac{[CH_{3}COONa]}{[CH_{3}COOH]} = 10^{pH - pKa} [/tex]
[tex] \frac{[CH_{3}COONa]}{[CH_{3}COOH]} = 10^{5.00 - 4.75} = 1.78 [/tex]
Now, the volume of the acetate solution is:
[tex]\frac{n_{CH_{3}COONa}/Vt}{n_{CH_{3}COOH}/Vt} = 1.78[/tex]
Since the total volume is the same, we have:
[tex]\frac{n_{CH_{3}COONa}}{n_{CH_{3}COOH}} = 1.78[/tex]
[tex] \frac{[CH_{3}COONa]_{i}*V_{b}}{[CH_{3}COOH]_{i}*Va} = 1.78 [/tex]
Solving for Vb
[tex] Vb = \frac{1.78*[CH_{3}COOH]_{i}*Va}{[CH_{3}COONa]_{i}} = \frac{1.78*1.00M*10.0mL}{1.00 M} = 17.8 mL [/tex]
Therefore, we need to add 17.8 mL of sodium acetate solution.
Find more here:
https://brainly.com/question/24188850?referrer=searchResults
I hope it helps you!
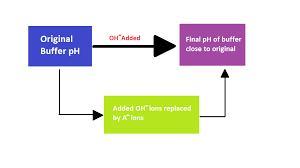
Related Questions
Which best describes the process that occurs when liquid water becomes
ice?
Answers
Answer:
Freezing
Explanation:
When a liquid goes to a solid, this process is called freezing.
Answer:
The water molecules get condensed and form a solid matter called ice. This process is called Condensation.
Kevin is working on a model that shows the positions of Earth, the Moon, and the Sun during the phases of
the Moon. How should he position them to show a New Moon?
Answers
Answer:
Explanation:what’s the answer
Answer: A. With Earth between the Moon and the Sun.
Explanation:
Hydrofluoric acid and Water react to form fluoride anion and hydronium cation, like this HF(aq) + H_2O(l) rightarrow F(aq) + H_3O^+ (aq) At a certain temperature, a chemist finds that a 5.6 L reaction vessel containing an aqueous solution of hydrofluoric acid, water, fluoride anion, and hydronium cation at equilibrium has the following composition: Calculate the value of the equilibrium constant K_C for this reaction. Round your answer to 2 significant digits. K_C =
Answers
Answer:
Kc = 1.09x10⁻⁴
Explanation:
HF = 1.62g
H₂O = 516g
F⁻ = 0.163g
H₃O⁺ = 0.110g
To solve this question we need to find the moles of each reactant in order to solve the molar concentration of each reactan and replacing in the Kc expression. For the reaction, the Kc is:
Kc = [H₃O⁺] [F⁻] / [HF]
Because Kc is defined as the ratio between concentrations of products over reactants powered to its reaction coefficient. Pure liquids as water are not taken into account in Kc expression:
[H₃O⁺] = 0.110g * (1mol /19.01g) = 0.00579moles / 5.6L = 1.03x10⁻³M
[F⁻] = 0.163g * (1mol /19.0g) = 0.00858moles / 5.6L = 1.53x10⁻³M
[HF] = 1.62g * (1mol /20g) = 0.081moles / 5.6L = 0.0145M
Kc = [1.03x10⁻³M] [1.53x10⁻³M] / [0.0145M]
Kc = 1.09x10⁻⁴Go to the Phase Changes tab. Select the particular molecule, and set the temperature to the given values by using the stove to increase or decrease the temperature. Increase the pressure of the cylinder by moving the lid of the cylinder down with the help of the finger. The pressure of the cylinder is indicated by the pressure gauge on the top left corner of the simulation. On the right-hand side of the simulation is the phase diagram for the molecule.
As the molecules collide on the walls of the cylinder, the pressure of the cylinder does not remain constant; it fluctuates slightly as indicated by the gauge.
In the phase diagram, observe the red point. When a particular molecule is selected, set the temperature and pressure condition to the required value. Observe the position of the red point on the phase diagram when the pressure reaches approximately near the required value.
Which of the following conditions will result in the formation of a supercritical fluid?
Check all that apply.
1. Oxygen at 100 K and no pressure
2. Argon at 160 K and approximately 6 atm pressure
3. Water at 294 K and approximately 1 atm pressure
4. Neon at 32 K and approximately 1 atm pressure
5. Neon at 50 K and approximately 10 atm pressure
6. Water at 894 K and approximately 20 atm pressure
7. Argon at 100 K and no pressure
8. Oxygen at 194 K and approximately 2 atm pressure
Answers
Answer:
2. Argon at 160K and approximately 6 atm pressure
5. Neon at 50K and approximately 10 atm pressure
6. Water at 894 K and approximately 20 atm pressure
8. Oxygen at 194K and approximately 2 atm pressure
Explanation:
Super critical fluid is formed when temperature rises beyond critical point. The state of molecules can be liquid or gas. The pressure should be above the certain level with an introduction of different gases at minimum of 2 atm pressure.
PLZ HELP URGENT!!!!!
Answers
Answer:
A fluke because it is an vertebrae
Explanation:
Limiting Reactant
12.0 grams of sodium reacts with 5.00 grams of chlorine. What mass of sodium
chloride could be produced?
Nas) +
Cl2(g) →
NaCls)
(1)
(2)
Identify the limiting reactant.
Determine the amount of sodium chloride produced.
Answers
Answer:
(1) Cl₂ is the limiting reactant.
(2) 8.18 g
Explanation:
2Na(s) + Cl₂(g) → 2NaCl(s)First we convert the given masses of reactants into moles, using their respective molar masses:
Na ⇒ 12.0 g ÷ 23 g/mol = 0.522 mol NaCl₂ ⇒ 5.00 g ÷ 70.9 g/mol = 0.070 mol Cl₂0.070 moles of Cl₂ would react completely with (2 * 0.070) 0.14 moles of Na. There are more Na moles than that, so Na is the reactant in excess while Cl₂ is the limiting reactant.
Then we calculate how many moles of NaCl are formed, using the limiting reactant:
0.070 mol Cl₂ * [tex]\frac{2molNaCl}{1molCl_2}[/tex] = 0.14 mol NaClFinally we convert NaCl moles into grams:
0.14 mol NaCl * 58.44 g/mol = 8.18 gWhich statement BEST describes why licensed health care workers are held to a higher standard?
A. They agree to ethical standards when they are licensed.
B. They agree to be good role models and always model good habits.
C. They are more involved than the public.
D. They are more educated than most of the public.
Answers
Answer:
They agree to ethical standards when they are licensed.
Explanation:
Answer:
C) They agree to ethical standards when they are licensed
Explanation:
What is the mass of 8.56 x 10^23 formula units of BaBr2? (3 sig figs in your answer)
Answers
296 g BaBr₂
General Formulas and Concepts:Math
Pre-Algebra
Order of Operations: BPEMDAS
Brackets Parenthesis Exponents Multiplication Division Addition Subtraction Left to RightChemistry
Atomic Structure
Reading a Periodic TableMolesAvogadro's Number - 6.022 × 10²³ atoms, molecules, formula units, etc.Stoichiometry
Using Dimensional AnalysisExplanation:Step 1: Define
[Given] 8.56 × 10²³ formula units BaBr₂
[Solve] grams BaBr₂
Step 2: Identify Conversions
Avogadro's Number
[PT] Molar Mass of Ba - 137.33 g/mol
[PT] Molar Mass of Br - 35.45 g/mol
Molar Mass of BaBr₂ - 137.33 + 2(35.45) = 208.23 g/mol
Step 3: Convert
[DA} Set up: [tex]\displaystyle 8.56 \cdot 10^{23} \ formula \ units \ BaBr_2(\frac{1 \ mol \ BaBr_2}{6.022 \cdot 10^{23} \ formula \ units \ BaBr_2})(\frac{208.23 \ g \ BaBr_2}{1 \ mol \ BaBr_2})[/tex][DA] Multiply/Divide [Cancel out units]: [tex]\displaystyle 295.99 \ g \ BaBr_2[/tex]Step 4: Check
Follow sig fig rules and round. We are given 3 sig figs.
295.99 g BaBr₂ ≈ 296 g BaBr₂
Thank you agenthammerx for helping me with this question!
Answer:
296 g BaBr₂
General Formulas and Concepts:
Math
Pre-Algebra
Order of Operations: BPEMDAS
Brackets
Parenthesis
Exponents
Multiplication
Division
Addition
Subtraction
Left to Right
Chemistry
Atomic Structure
Reading a Periodic Table
Moles
Avogadro's Number - 6.022 × 10²³ atoms, molecules, formula units, etc.
Stoichiometry
Using Dimensional Analysis
Explanation:
Step 1: Define
[Given] 8.56 × 10²³ formula units BaBr₂
[Solve] grams BaBr₂
Step 2: Identify Conversions
Avogadro's Number
[PT] Molar Mass of Ba - 137.33 g/mol
[PT] Molar Mass of Br - 35.45 g/mol
Molar Mass of BaBr₂ - 137.33 + 2(35.45) = 208.23 g/mol
Step 3: Convert
[DA} Set up:
[DA] Multiply/Divide [Cancel out units]:
Step 4: Check
Follow sig fig rules and round. We are given 3 sig figs.
295.99 g BaBr₂ ≈ 296 g BaBr₂
Rock is driven underground and changed by heat and pressure. This describes
what?
a. Igneous changing to sedimentary
b. Metamorphic changing to sedimentary
C. Sedimentary changing to metamorphic
d. Sedimentary changing to igneous
Answers
Answer:
Explanation:
metamorphic
An empty steel container is filled with 2.0 atm of H₂ and 1.0 atm of F₂. The system is allowed to reach equilibrium according to the reaction below. If Kp = 0.45 for this reaction, what is the equilibrium partial pressure of HF?
Answers
The equilibrium partial pressure of HF is 0.55 atm.
The equation of the reaction is;
H2(g) + F2(g) ⇄ 2HF
I 2 1 0
C -x -x +x
E 2 - x 1 - x x
We know that;
pH2 = 2.0 atm
PF2 = 1.0 atm
pHF = ??
Kp = 0.45
So;
Kp = (pHF)^2/pH2. pF2
0.45 = x^2/(2 - x) (1 - x)
0.45 = x^2/x^2 - 3x + 2
0.45(x^2 - 3x + 2) = x^2
0.45x^2 - 1.35x + 0.9 = x^2
0.55 x^2 + 1.35x - 0.9 = 0
x = 0.55 atm
Learn more about equilibrium: https://brainly.com/question/3980297
What is the concentration of a 22.35 L solution that contains 9.95 moles of sodium
acetate? Round your answer to the correct number of significant figures.
Answers
Answer:
0.445 M
Explanation:
Molarity = moles/Volume
M = 9.95/22.35 = 0.445 M
Please someone help me with this!!
Answers
Lewis dot diagram for the Cs1+ ion
Answers
Answer:
[tex]Cs^+[/tex]
Explanation:
Cesium Lewis dot structure would look like this:
·Cs, because it only has one valence electron. But, since it has a plus, that means we lost an electron. So, we have to get rid of the dot and you have:
[tex]Cs^+[/tex]
What are the details of the chemical structure of methylisothiazolinone (MIT)?
Answers
Ammonia can be made by reaction of water with magnesium nitride as shown by the following unbalanced equation: Mg3N2(s) + H2O(l) Mg(OH)2(s) + NH3(g) If this process is 71% efficient, what mass of ammonia can be prepared from 24.5 kg magnesium nitride?
Answers
The mass of ammonia prepared from 24.5 kg magnesium nitride, according to the reaction Mg₃N₂(s) + 6H₂O(l) → 3Mg(OH)₂(s) + 2NH₃(g), knowing that the process is 71% efficient is 5.87 kg.
The balanced reaction of production of ammonia is:
Mg₃N₂(s) + 6H₂O(l) → 3Mg(OH)₂(s) + 2NH₃(g) (1)
First, let's find the number of moles of magnesium nitride
[tex] n_{Mg_{3}N_{2}} = \frac{m_{Mg_{3}N_{2}}}{M_{Mg_{3}N_{2}}} [/tex] (2)
Where:
[tex]m_{Mg_{3}N_{2}}[/tex]: is the mass of Mg₃N₂ = 24.5 kg
[tex]M_{Mg_{3}N_{2}}[/tex]: is the molar mass of Mg₃N₂ = 100.9494 g/mol
The number of moles is (eq 2):
[tex] n_{Mg_{3}N_{2}} = \frac{m_{Mg_{3}N_{2}}}{M_{Mg_{3}N_{2}}} = \frac{24500 g}{100.9494 g/mol} = 242.70 \:moles [/tex]
We can calculate the mass of ammonia prepared, knowing that 1 mol of Mg₃N₂ reacts with 6 moles of H₂O to produce 3 moles of Mg(OH)₂ and 2 moles of NH₃ (reaction 1).
[tex]n_{NH_{3}} = \frac{2\: moles\: NH_{3}}{1\: mol\: Mg_{3}N_{2}}*n_{Mg_{3}N_{2} = \frac{2\: moles\: NH_{3}}{1\: mol\: Mg_{3}N_{2}}*242.70 \:moles \:Mg_{3}N_{2} = 485.4 \:moles[/tex]
Then, the mass of NH₃ is:
[tex] m_{NH_{3}} = n_{NH_{3}}*M_{NH_{3}} = 485.4 \:moles*17.031 g/mol = 8266.8 g = 8.27 kg [/tex]
Since the process is 71% efficient, the mass that can be prepared is:
[tex] m = 8.27 kg*0.71 = 5.87 kg [/tex]
Therefore, the mass of ammonia that can be prepared is 5.87 kg.
I hope it helps you!
The only three colors of light the human eye can detect are red, green, and blue.
O True
O False
Answers
what is the structure of methane
Answers
Answer:
CH4
Explanation:
if it is wrong, i blame my brain
A 50.0-mL volume of 0.15 M HBr is titrated with 0.25 M KOH. Calculate the pH after the addition of 13.0 mL of KOH. Express your answer numerically.
Answers
Answer:
pH= 1.17
Explanation:
The neutralization reaction between HBr (acid) and KOH (base) is given by the following equation:
HBr(aq) + KOH(aq) → KBr(aq) + H₂O(l)
According to this equation, 1 mol of HBr reacts with 1 mol of KOH. Then, the moles can be expressed as the product between the molarity of the acid/base solution (M) and the volume in liters (V). So, we calculate the moles of acid and base:
Acid:
M(HBr) = 0.15 M = 0.15 mol/L
V(HBr) = 50.0 mL x 1 L/1000 mL = 0.05 L
moles of HBr = M(HBr) x V(HBr) = 0.15 mol/L x 0.05 L = 7.5 x 10⁻³ moles HBr
Base:
M(KOH) = 0.25 M = 0.25 mol/L
V(HBr) = 13.0 mL x 1 L/1000 mL = 0.013 L
moles of HBr = M(HBr) x V(HBr) = 0.25 mol/L x 0.013 L = 3.25 x 10⁻³ moles KOH
Now, we have: 7.5 x 10⁻³ moles HBr > 3.25 x 10⁻³ moles KOH
HBr is a strong acid and KOH is a strong base, so they are completely dissociated in water: the acid produces H⁺ ions and the base produces OH⁻ ions. So, the difference between the moles of HBr and the moles of KOH is equal to the moles of remaining H⁺ ions after neutralization:
moles of H⁺ = 7.5 x 10⁻³ moles HBr - 3.25 x 10⁻³ moles KOH = 4.25 x 10⁻³ moles H⁺
From the definition of pH:
pH = -log [H⁺]
The concentration of H⁺ ions is calculated from the moles of H⁺ divided into the total volume:
total volume = V(HBr) + V(KOH) = 0.05 L + 0.013 L = 0.063 L
[H⁺] = (moles of H⁺)/(total volume) = 4.25 x 10⁻³ moles/0.063 L = 0.067 M
Finally, we calculate the pH after neutralization:
pH = -log [H⁺] = -log (0.067) = 1.17
milk milk milk........
Answers
Answer:
gimme that chocky milk.....
Explanation:
Answer:
m i l k
Explanation:
Which unit is NOT correctly paired?
O Liters : Volume
O Formula Mass : Count
O Grams: Mass
O Molecules : Mass
Answers
Answer:
Molecules : Mass
Explanation:
The first three answer choices correctly pair a unit with what it measures. Molecules are not used to measure mass; grams are.
Joan wants to test if salt lowers the temperature at which water boils. In two or more complete sentences, describe the best way for Joan to develop a hypothesis for this situation. Write your answer in the essay box below.
Answers
Answer:
Joan's hypothesis should make a prediction about the answer to the question. A hypothesis is just an educated guess on what you think the outcome will be on the experiment. The prediction must be testable and stated in if-then form. For example, a good hypothesis for Joan's experiment could be, I think that 1/2 cup of salt will make the water boil quicker, than water without salt.
Explanation:
Hope it helped!
Fill in the blanks. 3NH3
Answers
Answer:
3, 9, 3
Explanation:
The coefficient of 3 tells us that there are three molecules (the chemical unit of NH3). Each molecule of ammonia (NH3) is made up of 1 atom of nitrogen bonded to 3 atoms of hydrogen.
Since there are three molecules, we have three times the amount of atoms there are in one molecule.
3 x 1 = 3 nitrogen
3 x 3 = 9 hydrogen
Which of this is formed when a metal reacts with steam ? *
Carbon dioxide
A Salt
An oxide
A metal hydroxide
Answers
A metal hydroxide
I think so
The nucleus of an atom stays together only because the repulsive forces, called
forces, are overcome by
even stronger attractive forces.
fusion
nuclear
electrostatic
Answers
Answer:
Electrostatic
Explanation:
The forces that are overcome are the repulsive electrostatic forces between the protons (all charged positively).
How many molecules are in 450.0 grams of aluminum fluoride(AIF3)?
Answers
Answer:
It would be exactly 5.3586262014272155. But if you were to round it up it would be 5.35.
Use the solubility rules from the Lab 4 introduction and your knowledge of qualitative separation schemes from the lab to answer the following questions. The qualitative analysis experiment you did is actually an abbreviated version of a much larger analysis scheme in which many different cations are separated and identified. Suppose a mixture contains Ag , K , NH4 , Hg22 , Pb2 , Mg2 , Sr2 , Ba2 , Cu2 , Al3 and Fe3 .
(a) Which of the following ions could you separate, by causing them to precipitate, with the addition of HCl?
Ag+ K+ NH4+
Hg22+ Pb2+ Mg2+
Sr2+ Ba2+ Cu2+
Al3+ Fe3+
(b) After the addition of HCl, the above sample is centrifuged and decanted. Which of the following cations remaining in the supernatant could you separate, by causing them to precipitate, with the addition of H2SO4? (Hint: H2SO4 is a source of sulfate ions. Select all that apply.)
Ag+ K+ NH4+
Hg22+ Pb2+ Mg2+
Sr2+ Ba2+ Cu2+
Al3+ Fe3+
Answers
Answer:
a13+a13
Explanation:
In the UNBALANCED chemical reaction for the combustion of acetylene (used in welding torches), determine at standard temperature and pressure, how many liters of
H2O gas are produced if 12 liters of Oxygen gas are completely consumed?
__C2H2 + __O2 —> __CO2 + __H2O
(Please help! Random answers for points will be reported)
Answers
Answer:
2 C2H2 + 5 O2 = 4 CO2 + 2 H2O
I've checked this multiple times this should be it
Help me please tell me and I try to make u brainliest!
Answers
Which is a form of kinetic energy?
A. gravitational energy
B. chemical energy
C. electrical energy
D. sound energy
Answers
Hydrogen can be produced according to the following word equation
=zinc +hydrochloric acid =zinc chloride +hydrogen [zn^+2]
Write a complete balanced chemical equation for this chemical reaction?
Answers
Answer:
Zn + 2HCl → ZnCl₂ + H₂
Explanation:
First we write the equation using the molecular formulas instead of words:
Zn + HCl → ZnCl₂ + H₂We know zinc chloride is ZnCl₂ as the problem tells us the oxidation state of zinc in the products is +2, and chloride means Cl⁻¹.
Now we proceed to balance the reaction:
There are 2 Cl atoms and 2 H atoms on the right side, so we add a coefficient of 2 to HCl on the left side:
Zn + 2HCl → ZnCl₂ + H₂