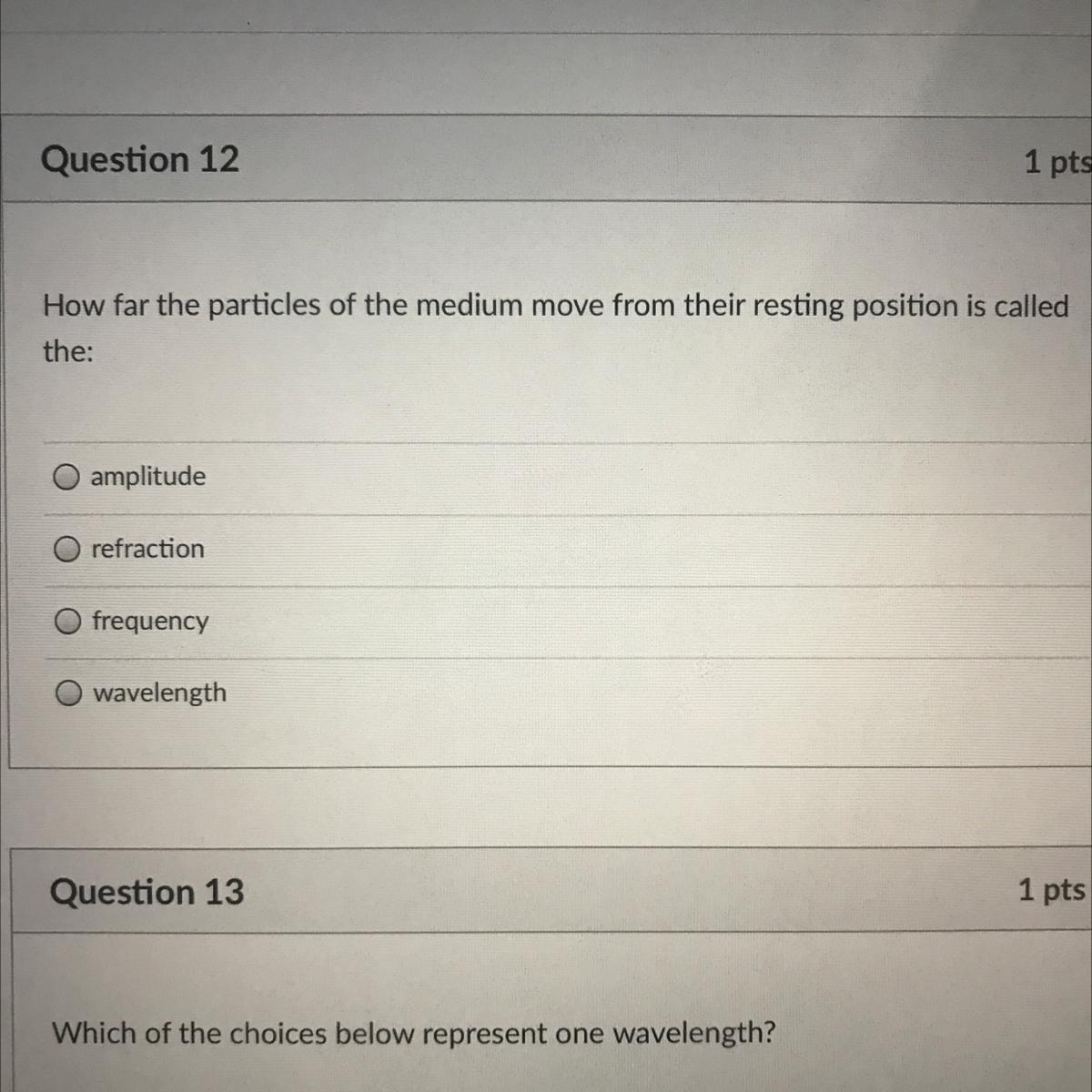
Answers
Answer:
Amplitude
Explanation:
Hope I helped :)
Related Questions
What is produced when concentrated aqueous sodium chloride is electrolysed?
Answers
You’re welcome!! Have a good day/night :)
A certain protein transports a sodium ion from the inside of the cell membrane to the outside using active transport. Which of the following must be true? A. There is no change in concentration before or after transport. B. The transport process did not use chemical energy. C. After transport, the concentration of protein in the cell decreases. D. After transport, the concentration of sodium is lower inside the cell.
Answers
Which statement best describes the energy in the food web? A. The amount of energy available at level B depends on the amount of energy available at level C. B. The amount of energy available at level A depends on the energy provided from an abiotic source. C. The amount of energy available at level C is greater that the amount of energy available ate level A. D. The amount of energy available at level B is transferred to level A
Answers
Answer:
The amount of energy available at level A depend on energy provided from an a biotic source
1) How many molecules are in 0.02 moles of beryllium iodide?
Answers
Answer:
3 grams??
Explanation:
I think this would be the answer you are looking for.
How are fossil fuels formed from geologic processes?
Answers
Answer:
Explanation:
Fossil fuels are hydrocarbons, namely, petroleum , coal and natural gas, formed from buried combustible geologic deposits of organic and natural materials. They are created from decayed animals and plants that convert into coal, crude oil or heavy oil due to constant exposure to pressure and heart in the earth’s crust.
The compounds that make up plankton and plants convert into fossil fuels. Plankton decomposes into natural gas and oil, while plants become coal. Nowadays, humans extract these resources through coal mining and the drilling of oil.
What are fossil fuel ?The term fossil fuel is defined as they are made from decomposing plants and animals. An example of a fossil fuel is coal, as well as oil and natural gas.
Because they were generated from the petrified, buried remains of plants and animals that once lived millions of years ago, coal, crude oil, and natural gas are all referred to as fossil fuels.
Thus, The remains of these animals underwent a transformation into what we now know as fossil fuels over millions of years at great pressure and high temperature.
To learn more about the fossil fuel, follow the link;
https://brainly.com/question/2029072
#SPJ2
Which system is affected by the process of weathering?
the hydrosphere
the atmosphere
the biosphere
the geosphere
Answers
[tex] \huge \colorbox{pink}{Answer☟︎︎︎}[/tex]
When ice melts, liquid water performs the act of erosion by carrying away the tiny rock fragments lost in the split. This specific process (the freeze-thaw cycle) is called frost weathering or cryofracturing. Temperature changes can also contribute to mechanical weathering in a process called thermal stress.
At room temperature, oxygen is a gas and hydrogen is a gas. When they combine at room temperature, they form a _______________.
Answers
Answer: they can combine to form water or hydrogen peroxide.
Explanation:
sorry if it wrong i just looked it up lol. hope this helps tho
Copper has a specific heat of 0,900 J(g C). How much energy in kJ is needed to raise the temperature of a 700 g block of aluminum from 30.7°C to 82,1C?(show your steps)
Answers
Answer:
32.4 kJ
Explanation:
Given that:
Specific heat capacity fo copper, c = 0.9
Mass of copper, m = 700 g
Temperature change, dθ ;t2 - t1 = (82.1 - 30.7) = 51.4
Energy required, E = mcdθ
E = 700 * 0.9 * 51.4
E = 32382 J
Energy required on kJ
32382 / 1000
= 32.382 kJ
= 32.4 kJ
Convert 23.92 mm to meter
Answers
Answer:
0.02392 m
Explanation:
Kilo - Hecto - Deka - Meters/Liters/Grams - Deci - Centi - Milli
23.92 mm ---> 0.02392 m
Basically move the decimal 3 units to the left.
Hope this helps and stay safe, happy, and healthy, thank you :) !!
"A bicycle's brakes apply 299 N of frictional force to the wheels as the bike moves 24 m
for 3.0 s. How much WORK do the brakes do?"
Answers
Answer:
0.62 N
Explanation:
first of all: 3.0 s = 0.05 m
Let ? be the work that brakes do at 3.0s
24 m ----> 299 N
0.05 m ---->?
? = (0.05 m x 299 N)/ 24
? = (14.95)/ 24
? = 0.622 N
A molecule is the smallest part of a ?
Answers
Answer:
A molecule is the smallest particle in a chemical element or compound that has the chemical properties of that element or compound. Molecules are made up of atoms that are held together by chemical bonds. These bonds form as a result of the sharing or exchange of electrons among atoms.
Explanation:
Answer:
A molecule is the smallest part of an element, sorry if im incorrect
The iodide (I2) content of a commercial mineral water was measured by two methods that produced wildly different results.6 Method A found 0.23 milligrams of I2 per liter (mg/L) and method B found 0.009 mg/L. When Mn21 was added to the water, the I2 content found by method A increased each time that more Mn21 was added, but results from method B were unchanged. Which of the Terms to Understand describes what is occurring in these measure- ments? Which result is more reliable?
Answers
Answer:
Results from method B is more reliable than method A.
Explanation:
The two method that are used for the analysis produced different results. The first method that is method A gives higher value of the iodine content than the method B.
When [tex]$Mn^{2+}$[/tex] was added to water, method A showed an increased in the iodine content and it increases with the increase in the amount of [tex]$Mn^{2+}$[/tex] .
Where as in the method B, there is no change in the results. Therefore the measurements provided by the method A shows an inference of [tex]$Mn^{2+}$[/tex] ion.
The measurement of the iodine content is affected by the presence of the ion [tex]$Mn^{2+}$[/tex] in water.
Since in method B there is no change in measurement, it is independent of the presence [tex]$Mn^{2+}$[/tex] ion in water.
As higher iodine content is given by method A, so [tex]$Mn^{2+}$[/tex] ion must be present in original water that must be interfering the measurement. Hence, method B is more reliable.
Method B is more reliable as its value is not altered by external factors such as contamination.
What is water analysis?Water analysis is done to test the quality of water.
The method employed in water analysis is titrimetric analysis.
In the test iodide (I⁻) content of a commercial mineral water using the two methods, the results of the test showed different values for the iodide content.
However, since addition of Mn²⁺ to the mineral water increases the iodide content value from Method A while that of Method B remains unchanged, Method B is more reliable as its value is not altered by external factors such as contamination.
Learn more about titrimetric analysis at: https://brainly.com/question/8463543
HELP I WILL GIVE YOU BRAINLIST!!!
Pick one substance (ex: water and table salt) and compare two of the atoms connected to each other (ex: H and O, Na and Cl) and answer the following questions for each pair:
1)Are they close or far away from each other on the periodic table?
Do they have similar atomic radii?
2)Do they have a large or small difference in ionization energy? What about for electron affinity?
Answers
Help me with my homework plzy
Answers
Answer:
the second answer choice i think
Explanation:
thermal energy is what we call energy that come from ___ of matter
Answers
2 CH3OH + 3 02 2 CO2 + 4H2O
What is the mass of oxygen (O2) that is required to produce 579 g of carbon dioxide (CO2)?
. Your answer should have three significant figures.
Answers
Answer:
632 g
Explanation:
2CH₃OH + 3O₂ → 2CO₂ + 4H₂OFirst we convert 579 g of CO₂ into moles, using its molar mass:
579 g CO₂ ÷ 44 g/mol = 13.16 mol CO₂Then we convert CO₂ moles into O₂ moles, using the stoichiometric coefficients:
13.16 mol CO₂ * [tex]\frac{3molO_2}{2molCO_2}[/tex] = 19.74 mol O₂Finally we convert O₂ moles into grams, using its molar mass:
19.74 mol O₂ * 32 g/mol = 632 gI need help with this chem problem!
Answers
what makes a substance a good insulator
Answers
Answer:
zoo wee mama
Explanation:
The particles that make up an object are held together by bonds. Insulators have strong bonds that hold their particles rigidly in place. This prevents particles from gaining energy and increasing the temperature. Wool, dry air, plastics, and polystyrene foam are all examples of good insulators.
State the coefficient required to correctly balance the following chemical equation:
_____ Ca3(PO4)2+ _____ NaCl ---------> _____ Na3PO4 + _____ CaCl2
Answers
Answer:
1 Ca3(PO4)2+ 6 NaCl ---------> 2 Na3PO4 + 3 CaCl2
The percent composition by mass of nitrogen in NH OH (gram-formula mass = 35 grams/mole) is equal to
Answers
Answer:
the percentage composition of mass of nitrogen in NH OH is 42.86 %
please help me
is for my daughter
Answers
Answer:
I think you have to freeze the water itself then take the temp with a thermometer ️of each one then record how long it takes each one to start freezing then how long it takes for it to melt
Explanation:
sorry if I'm wrong but you have to do the experiment yourself then fill it out
How many grams equal 4.3 x 1024 atoms of oxygen (02)?
Answers
Answer: 248.66g
Explanation:
4.3e24 / 6.23e23 = 6.9 mols O2
6.9(18.02*2)=248.66
how many significant digits are in the number 1.0 × 10^3?
Answers
1•10^3 add three 0’s
Hydrogen has an oxidation number of +1 in all compounds excepts when it binds with metals and has an oxidation state of -1.
Answers
Answer:
See explanation
Explanation:
In the periodic table, hydrogen is placed in group 1 for convenience. Hydrogen is not a metal, however, its common oxidation state is +1. It easily forms a univalent positive ion just like group 1 elements.
So, when hydrogen is combined with strongly electropositive metals such as sodium, lithium, etc, hydrogen accepts one electron and exhibits an oxidation state of -1 in those compounds.
Such compounds in which hydrogen is combined with strongly electropositive metals to give a binary compound is called a hydride.
PLEASE PLEASE I do not understand may someone kindly help me pls
Answers
Answer:
It can be verified since the information came from a government article. Or you could trust the magzine/general article if its from a well known article. Explanation:
you get the point, you can add on to this. I can't make it the best since im not in your class learning. Good luck
CAN SOMEONE PLEASE HELP ME I WILL MARK AS BRAINLIEST :(
Answers
Answer:
ok so i did the problem and the answer was 8e+29
Explanation:
i dont really know what the answer is but thats the answer i got
sorry if this didnt help
molar mass of Mg(CO3)
Answers
Answer:
[tex]\boxed {\boxed {\sf 84.313 \ g/mol}}[/tex]
Explanation:
The molar mass is the mass per mole of an element or substance. The values for atomic mass on the Periodic Table are used as the molar mass. Look up the molar masses for the elements in the formula.
Mg: 24.305 g/molC: 12.011 g/molO: 15.999 g/molAnalyze the formula. Magnesium and carbon have no subscript, so there is only one atom of each. Oxygen has a subscript of 3, so there are 3 atoms and the mass has to be multiplied by 3.
MgCO₃= 24.305 + 12.011+ 3(15.999) MgCO₃= 24.305+12.011+47.997=84.313 g/molThe molar mass of magnesium carbonate is 84.313 grams per mole.
One component of smog is nitrogen
monoxide, NO. A car produces about 8 g of
this gas per day. What is the volume at STP?
Answers
Answer:
V = 5.97 L
Explanation:
Hello!
In this case, according to the ideal gas law:
[tex]PV=nRT[/tex]
We would need to solve for V as we know the temperature and pressure as the gas it at STP conditions (273 K and 1 atm respectively):
[tex]V=\frac{nRT}{P}[/tex]
Next, we compute the moles in 8 g of NO, given its molar mass of 30.01 g/mol:
[tex]n=\frac{8g}{30.01g/mol}=0.267mol[/tex]
Therefore, we obtain the following volume:
[tex]V=\frac{0.267mol*0.08206\frac{atm*L}{mol*K}*273K}{1atm}\\\\V=5.97 L[/tex]
Best regards!
A group of students is setting up a test to see whether earthworms, like those shown in the photo, prefer rough or smooth surfaces
Answers
Answer:
they like smooth
Explanation:
When a hydrocarbon fuel is burned, almost all of the carbon in the fuel burns completely to form CO2 (carbon dioxide), which is the principal gas causing the greenhouse 102 ENERGY, ENERGY TRANSFER effect and thus global climate change. On average, 0.59 kg of CO2 is produced for each kWh of electricity generated from a power plant that burns natural gas. A typical new household refrigerator uses about 700 kWh of electricity per year. Determine the amount of CO2 production that is due to the refrigerators in a city with 300,000 households.
Answers
Answer:
1.239 * 10^8 Kg
Explanation:
Since all the electricity consumed comes from natural gas;
amount of electricity consumed = 700 * 300,000 = 2.1 * 10^8 kWh
So, amount of CO2 consumed is given by;
amount of electricity consumed * amount of CO2 per kWh
Hence,
Amount of CO2 = 2.1 * 10^8 kWh * 0.59 = 1.239 * 10^8 Kg