Answers
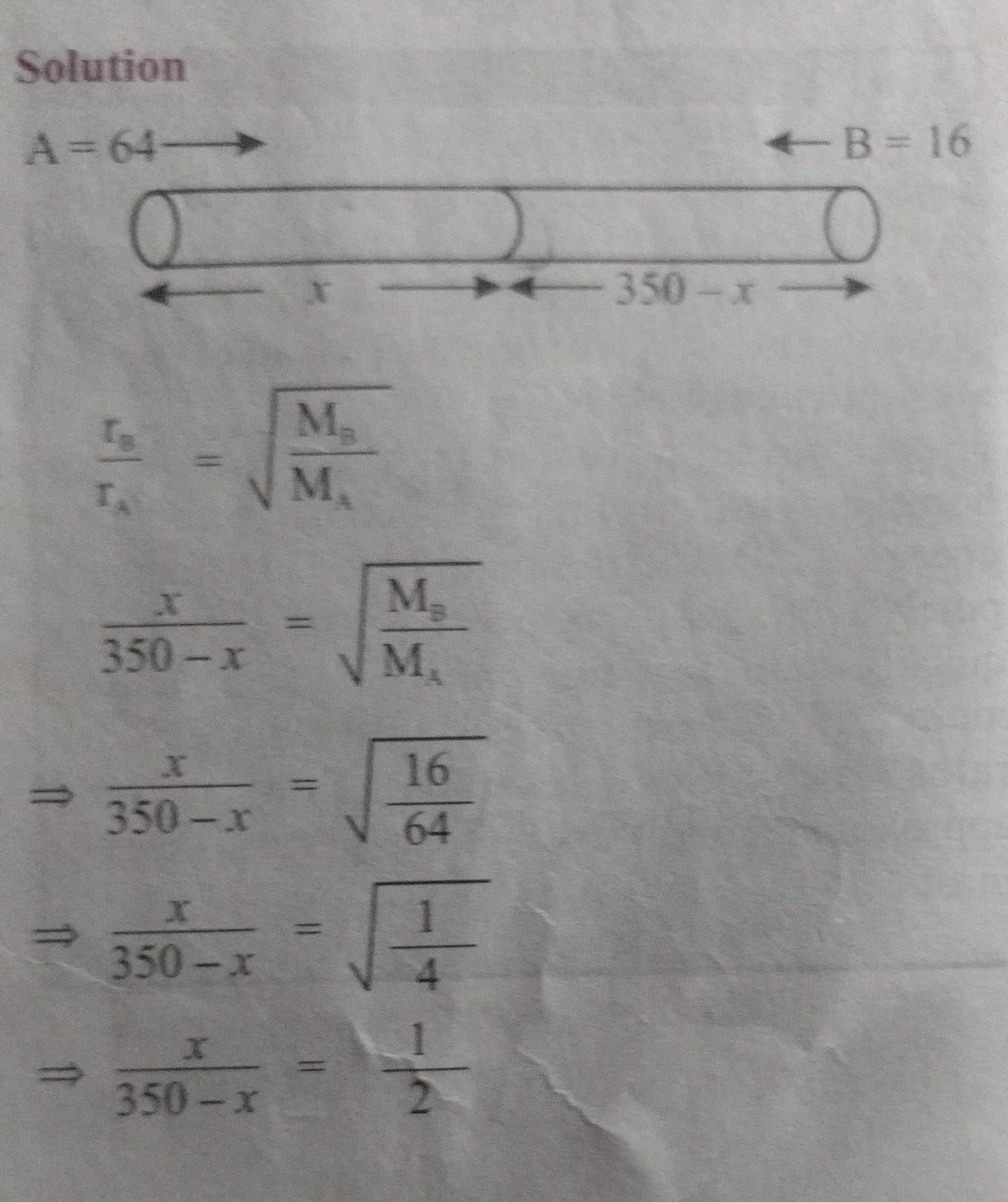
2x = 350- x
3x = 350
x = 350/x
therefore,
x = 116.67cm.
so, (350 - x)cm³ = (350- 166.67)cm³ = 233.33cm³
Hence distance covered by gas A is 116.67cm that by B is 233.33cm
Related Questions
If the density of aluminum is 2.7g/ml, what is the volume of 12.0g?
Answers
Answer:
The answer is 4.44 mLExplanation:
The volume of a substance when given the density and mass can be found by using the formula
[tex]volume = \frac{mass}{density} \\ [/tex]
From the question we have
[tex]volume = \frac{12}{2.7} \\ = 4.4444444....[/tex]
We have the final answer as
4.44 mLHope this helps you
Consider the balanced equation below. Upper P Upper C l Subscript 3 Baseline + Upper C l Subscript 2 Baseline right arrow Upper P Upper C l Subscript 5. What is the mole ratio of PCl3 to PCl5? 1:1 2:1 3:5 5:3
Answers
Answer : The mole ratio of [tex]PCl_3[/tex] to [tex]PCl_5[/tex] is 1 : 1.
Explanation :
Balanced chemical reaction : It is a chemical reaction in which the number of atoms of individual elements present on reactant side must be equal to the product side.
The balanced chemical reaction is:
[tex]PCl_3+Cl_2\rightarrow PCl_5[/tex]
By the stoichiometry of the reaction we can say that 1 mole of [tex]PCl_3[/tex] reacts with 1 mole of [tex]Cl_2[/tex] to give 1 mole of [tex]PCl_5[/tex].
From this we conclude that the mole ratio of [tex]PCl_3[/tex] to [tex]PCl_5[/tex] is 1 : 1.
Hence, the mole ratio of [tex]PCl_3[/tex] to [tex]PCl_5[/tex] is 1 : 1.
Answer:
A
Explanation:
20
How do you determine the number of barium atoms in the formula below?
4Ba(OH)2
F
# of Ba atoms = coefficient 4 + subscript 1 = 5
G
# of Ba atoms = coefficient 4 X subscript 1 = 4
H
# of Ba atoms = subscript 4 X coefficient 2 = 6
# of Ba atoms coefficient 1 + subscript 1 = 2
J
Answers
Answer:
G is correct option:
# of Ba atoms = coefficient 4 × subscript 1= 4
Explanation:
Formula:
4Ba(OH)₂
G is correct option:
# of Ba atoms = coefficient 4 × subscript 1= 4
Because there are only 4 atoms of Ba in given formula.
Ba(OH)₂ contain one atom of Ba while in question there are 4 moles of Ba(OH)₂ present thus total 4×1 = 4 atoms of Ba present.
Other options are incorrect. Because,
F:
# of Ba atoms = coefficient 4 + subscript 1 = 5
This shows given formula contain 5 Ba atoms. So it is incorrect.
H:
# of Ba atoms = subscript 4 × coefficient 2 = 6
This shows that subscript is 4 which is incorrect because coefficient is 4 and subscript is 1.
j:
# of Ba atoms = subscript 1 + coefficient 1 = 2
This option shows that subscript is one which is correct but coefficient is incorrect. The coefficient of Ba is 4 and coefficient is always multiply with subscript not added. So this option is also incorrect.
A compound containing only carbon, hydrogen, and oxygen is analyzed using combustion analysis. When 50.1 g of the compound is burned, 91.8 g of carbon dioxide and 25.1 g of water are collected. In order to determine the moles of carbon in the compound, first determine the moles of carbon dioxide that were produced from the combustion.
Answers
Answer:
[tex]C_{3}H_4O_2[/tex]
Explanation:
Hello,
In this case, since the carbon of the initial compound is present in the carbon dioxide product, we can compute the mass and moles of carbon in the compound:
[tex]n_C=91.8g CO_2*\frac{1molCO_2}{44gCO_2} *\frac{1molC}{1molCO_2} =2.09molC\\\\m_C=91.8g CO_2*\frac{1molCO_2}{44gCO_2} *\frac{1molC}{1molCO_2}*\frac{12gC}{1molC} =25.0gC[/tex]
Next, the mass and moles of hydrogen in the compound, is contained in the yielded amount of water, thus, we compute the mass and moles of hydrogen in the compound:
[tex]n_H=25.1gH_2O*\frac{1molH_2O}{18gH_2O} *\frac{2molH}{1molH_2O} =2.79molH\\\\m_H=25.1gH_2O*\frac{1molH_2O}{18gH_2O} *\frac{2molH}{1molH_2O} *\frac{1gH}{1molH} =2.79gH[/tex]
In such a way, the mass of oxygen comes from the mass of the compound minus the mass of carbon and oxygen:
[tex]m_O=50.1g-25.0g-2.79g=22.31gO[/tex]
And the moles:
[tex]n_O=22.31gO*\frac{1molO}{16gO}=1.39molO[/tex]
Then, we compute the subscripts by diving the moles of C, H and O by the moles of oxygen as the smallest moles:
[tex]C:\frac{2.09}{1.39}=1.5 \\\\H:\frac{2.79}{1.39}=2\\ \\O:\frac{1.39}{1.39} =1[/tex]
After that, we write:
[tex]C_{1.5}H_2O[/tex]
Which must be shown in whole number only, thereby we multiply the subscripts by 2, so the empirical formula turns out:
[tex]C_{3}H_4O_2[/tex]
Best regards.
what's the summary of matter
Answers
matter is anything that has mass and volume...it has three state thus solid, liquid and gas.. It possess chemical and physical changes.
How many moles are in 141.16 grams of F?
Use two digits past the decimal for all values
Answers
Answer: 2681.81
Explanation:
hope that helps
Please help me!!!:)))
Answers
Answer:
blocks 1 and 2 the rhdh huff hgfhh5
g Calculate the mass percent of sodium bicarbonate in the solution that has 7.00 g of solution and 0.052 Kg of water.
Answers
Answer:
11.86%
Explanation:
First, we convert both solvent and solute to the same unit, say, kg. We have.
Mass of Sodium Bicarbonate = 7g = 7/1000 kg = 0.007 kg
Mass of water = 0.052 kg.
Formula for the mass percent is
% of sodium bicarbonate = [(mass of sodium bicarbonate) / (mass of total solution) * 100%]
Total mass of solution = 0.007 + 0.052 Total mass of solution = 0.059
% of sodium bicarbonate = 0.007 / 0.059 * 100%
% of sodium bicarbonate = 11.86%
Therefore, the mass percent of sodium bicarbonate I'm the solution is 11.86%
If it takes 26.0 mL of 0.0250 M potassium dichromate to titrate 25.0 mL of a solution containing Fe2 , what is the molar concentration of Fe2
Answers
Answer:
Explanation:
moles of potassium dichromate = .0250 x .026 = 65 x 10⁻⁵ moles
1 mole of potassium dichromate reacts with 6 moles of Fe⁺²
65 x 10⁻⁵ moles of potassium dichromate will react with
6 x 65 x 10⁻⁵ moles of Fe⁺²
= 390 x 10⁻⁵ moles
390 x 10⁻⁵ moles are contained in 25 mL of solution
molarity of solution = 390 x 10⁻⁵ / 25 x 10⁻³
= 15.6 x 10⁻² M .
How many moles would be in 24.23 grams of SrSO4?
Use two digits past the decimal for all values.
Answers
Answer:
about 0.13 mol
Explanation:
To find number of mols when given grams you first have to find the molar mass of the compound. This is done by adding up the atomic masses of the element in the compound. So Sr= 88 g/mol S=32 g/mol and O=16 g/mol. Then 88+32+(16x4)=184. Then using this you can convert from grams to mols by dividing the grams by the molar mass. So, 24.23/184 equals about 0.13 mol.
When a helium balloon rises in the air, it expands. If the volume of the balloon doubles, what happens to the density of the helium inside it?
a.The density decreases by half
b.The density doubles
c.The density triples
d.The density stays the same
Answers
7. The equation below represents a chemical reaction that occurs in living cells.
CH,26 + 602
6CO2 + 6H,0 + energy
How many atoms are represented in the reactants of this equation?
A=6
B=12
C=24
D=36
Answers
Answer:
D
Explanation:
Because i just added all of numbers up
C. 24 if the zero after the 6H, is supposed to be a zero however if it wasnt and u were trying to say h2o it'd be 36
Given the mass of carbon on the periodic table (12.01 amu), which isotope do you think is the most abundant? Explain WHY in complete sentences. *
Answers
Answer:
Carbon-12
Explanation:
Carbon-12 is a carbon with 6 protons and 6 neutrons. Since the AMU for an element is calculated by averaging all naturally occurring isotopes, we can see that 12 in the most abundant. (Since 12.01 is very close to 12, the only reason it is not exactly 12 is because there are other isotopes like carbon-13 or carbon-14 but in low amounts)
What is the atomic number of arsenic (As)?
O A. 33
O B. 15
C. 75
D. 4
SUB
Answers
Answer:
33
Explanation:
Ok
A plant part that contains a tiny new plant, or embryo
seed
spore
root
flower
Answers
how many moles of h2 can be made from the complete reaction of 3.5 moles of al?
Given: 2Al+6HCL 2Alcl3+3h2
Answers
Answer:
From the given equation, we can see that for every 2 moles of Al, we get 3 moles of H2
So, we can say the the number of moles of H2 is 3/2 times the number of moles of Al
We are given the number of moles of Al and we have to find the number of moles of H2
We have deduced the relationship:
Moles of Al * 3 / 2 = Moles of H2
Replacing the variables with given values
3.5 * 3 / 2 = Moles of H2
Moles of H2 = 5.25 moles
Which is denser a liquid or solid why?
Answers
Answer:
Liquids are usually less dense than solids but more dense than air. Temperature can change a liquid's density. For example, increasing the temperature of water causes the molecules to spread farther apart. The farther apart the molecules are, the less dense the water is.
Answer:
Solids are usually much more dense than liquids and gases, but not always.
Explanation:
Mercury, a metallic element that is a liquid at room temperature, is denser than many solids. Aerogel, a very unusual human-made solid, is about 500 times less dense than wate
The reaction between HCl and KOH results in an increase in temperature in the solution. Select the correct statement from the list below.
a) this is an endothermic reaction
b) this is a phase change reaction
c) this is a vaporization reaction
d) this is an exothermic reaction
Answers
Answer:
d) this is an exothermic reaction.
Explanation:
The reaction between HCl and KOH results in an increase in temperature in the solution. Select the correct statement from the list below.
a) this is an endothermic reaction . NO. This would cause a decrease in the temperature of the solution.
b) this is a phase change reaction . NO. All the species remain in the aqueous phase.
c) this is a vaporization reaction . NO. All the species remain in the aqueous phase.
d) this is an exothermic reaction. YES. The reaction releases heat, so it is exothermic.
Which element is classified as a noble gas?
Answers
Answer:
The elements with completely filled shells are classified as noble gases
That is why we only see noble gases on the rightmost corner of the periodic table, it is because they have the maximum number of electrons in a shell
Examples of noble gases
Helium , Neon , Argon and Krypton are some examples of noble gases
Answer:
D. (Xe) XenonExplanation:
I JUST TOOK THE TEST!
A solution has a pH of 6. What is true about the solution?
A. It is a strong basic solution.
B. It is a weak acidic solution.
C. It is a weak basic solution.
D. It is a strong acidic solution.
please help me
Answers
Answer:
A. it is a strong basic solution
Answer:
(see below)
Explanation:
First, refer to the pH scale:
1 2 3 4 5 6 7 8 9 10 11 12 13 14
<== acidic neutral basic ==>
You can see that the smaller the number, the stronger the acid and the bigger the number, the more basic the base is. 7 is neutral, such as water. it's neither basic nor acidic.
Now, using the process of elimination:
A) It's a strong basic solution.
No, because this solution's pH hasn't even reached basic.
B) It's a weak acidic solution.
Yes, because it is acidic and it's just a little bit more acidic than a neutral solution.
C) It's a weak basic solution.
No, because this solution's pH hasn't even reached basic.
D) It's a strong acidic solution.
No, because even though it's acidic, it's just below neutral. For something to be a strong acidic solution would be around a pH of 3.
So the answer would be B) It's a weak acidic solution.
Calculate the number of moles in 2.60 grams of SnO2 ? Please show your work to receive credit.
Answers
Answer:
There are 0.017252 moles in SnO2
Explanation:
n= m/M
n=2.6/(118.710 + 2(16))
n= 2.6/150.71
n= 0.017252
Remeber to round based on the significant digits in the question.
An alkali metal with fewer than 10 protons in its nucleus
Answers
Answer:
Lithium (Li)
Explanation:
It has an atomic number of 3.
ppm is used to describe very small amounts of substance?
Answers
parts per million (ppm), parts per billion (ppb) and parts per trillion (ppt) are the most commonly used terms to describe very small amounts of substances. a ppm of a chemical in water means that, in a million units of water, there would only be one unit of the chemical.
You are given 3 liquid samples – A, B, C and told that one is water, one is ethanol, and one is ether (d=0.700 g/mL). Each sample is 10.0 mL. the masses are as follows: A = 10.0 g, B= 7.00 g, C = 8.00 g. Identify each of the substances using density problems.
Answers
Answer:
im just trying to get work done i will help u if i knew it
2NH.
N2 + 3H2
Reactants
Product
On the balanced equation above, how many
atoms of each element are in the reactant?
N =
H =
Answers
Answer:
N=2
H=6
Explanation:
1.Balance a chemical equation in terms of moles.
2.Use the balanced equation to construct conversion factors in terms of moles.
3.Calculate moles of one substance from moles of another substance using a balanced chemical equation.
The law of conservation of matter says that matter cannot be created or destroyed. In chemical equations, the number of atoms of each element in the reactants must be the same as the number of atoms of each element in the products.
(P.s it could also be where you have to solve it in which you have to simplify it first then solve it.) like adding them all up.
Hope this is the answer. :)
What is the mass in grams of 2.4 x 10-2 mol K2SO4
Answers
4.18 grams
Hope it helps!
Based on the visible cell structures, which of the following statements is true?
All of the cells are plant cells.
All of the cells have chloroplasts.
All of the cells are animal cells.
All of the cells have a nucleus.
Answers
State Hess' law of constant heat summation.
(b) Calculate the enthalpy of formation of CH4 from the following data:
i) C(s) + O2(g) → CO2(g); ∆H = -393.7 kJ/mol
ii) H2(g) + 1⁄2 O2(g) → H2O(l); ∆H = -285.8 kJ/mol
iii) CH4(g) + 2 O2(g)→ CO2(g) + 2H2O(l); ∆H = -890.4 kJ/mol
Answers
Answer:
-74.6 kj/mol
Explanation:
you can see the answer at the pic
The heat of fusion for naphthalene is 0.148 kJ/g. Calculate the heat energy in joules required to melt 52.95 g of naphthalene.
Answers
Answer:
7836.6 J
Explanation:
The following data were obtained from the question:
Heat of fusion (Hf) = 0.148 KJ/g
Mass (m) = 52.95 g
Heat (Q) required =..?
The heat (Q) required to melt the sample of nephthalene can be obtained as follow:
Q = m·Hf
Q = 52.95 × 0.148
Q = 7.8366 KJ
Finally, we shall convert 7.8366 KJ to Joule (J) in order to obtain the desired result. This can be obtained as follow:
1 kJ = 1000 J
Therefore,
7.8366 KJ = 7.8366 KJ × 1000 J / 1 KJ
7.8366 KJ = 7836.6 J
Therefore, 7.8366 KJ is equivalent to 7836.6 J
Thus, 7836.6 J of heat energy is required to melt the sample of nephthalene.
The heat required for melting of 52.95 g of naphthalene has been 7.8366 kJ.
The heat of fusion has been the energy required by 1g for the change in the substance.
The heat required to melt naphthalene can be given as:
Heat = mass × Heat of fusion
The given mass = 52.95 g
The heat of fusion = 0.148 kJ/g
The heat required can be given as:
Heat = 0.148 × 52.95 kJ
Heat = 7.8366 kJ.
The heat required for melting of 52.95 g of naphthalene has been 7.8366 kJ.
For more information about the heat of fusion, refer to the link:
https://brainly.com/question/25475410
What is the mass in grams of 5.9 mol c8 h18
Answers
Answer:
mass = density × volume = 0.67 × 1.00 = 0.67 kg = 670 g. The molar mass of octane, C8H18, is 8 × 12.01 (C) + 18 × 1.008 (H) = 114.224.
Explanation: