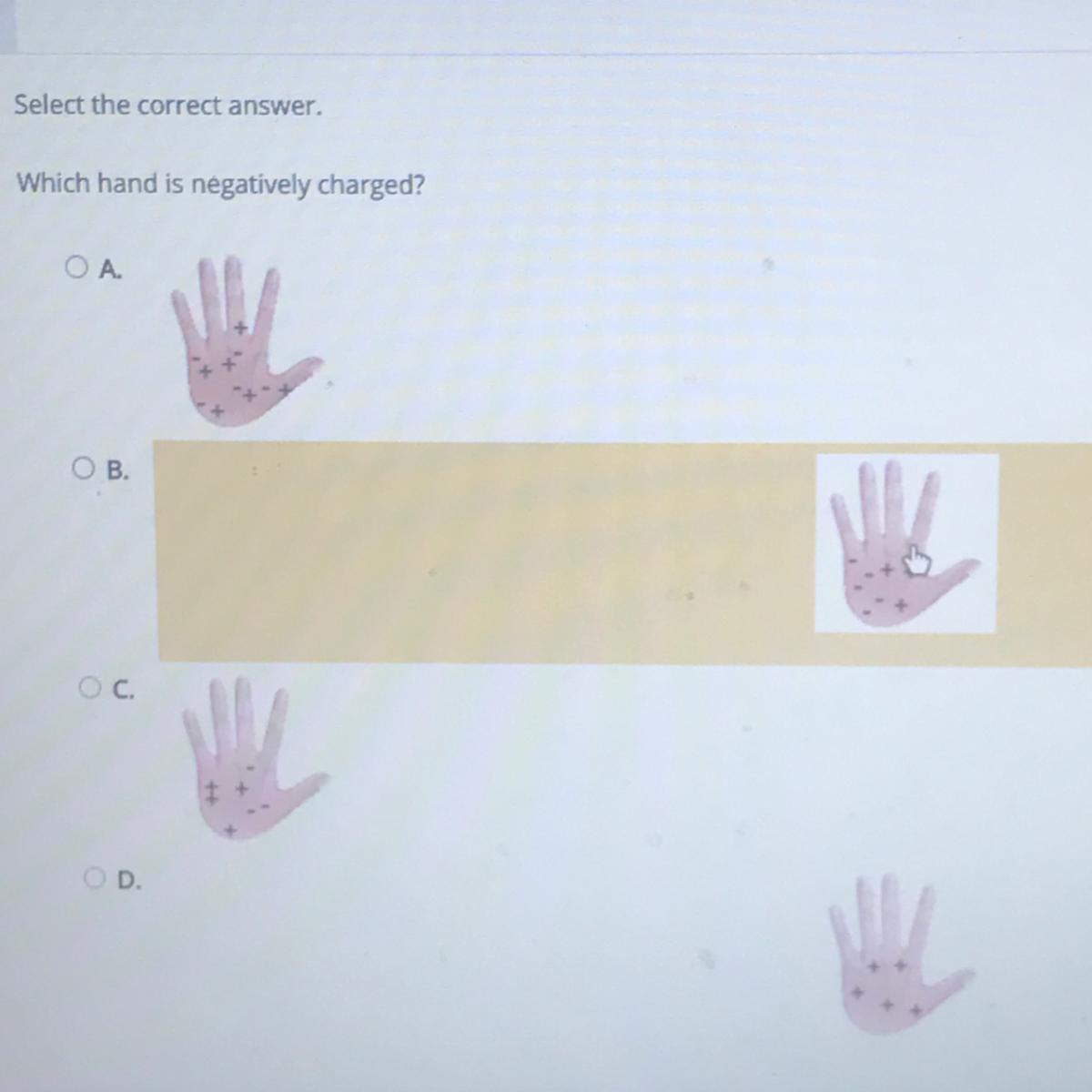
Answers
Answer:
I think B
Explanation:
There are more negative ions than positive ions
Related Questions
Really stuck on this question !! Pls help
Answers
Answer:
can not be determined
Explanation:
because when I tried to calculate it it didn't give me the answer options that was in the pic so it's definitely C
PLEASE PLEASE HELP!!!!
Approximately what mass of potassium nitrate will eventually precipitate from a supersaturated solution containing 19 grams of the solute in 28 grams of water at 40C°?
Answers
A supersaturated solution contains more solute at a given temperature than is needed to form a saturated solution.
Increased temperature usually increases the solubility of solids in liquids.
For example, the solubility of glucose at 25 °C is 91 g/100 mL of water. The solubility at 50 °C is 244 g/100 mL of water.
If we add 100 g of glucose to 100 mL water at 25 °C, 91 g dissolve. Nine grams of solid remain on the bottom. We have a saturated solution.
Hope it helps u
Plz mrk me brainlest
Commercial soaps are mixtures of ionic compounds typically made up of monatomic cations, such as Na and K , and organic polyatomic anions derived from fatty acids. These negatively charged molecular ions are characterized by the presence of hydrocarbon chains which are 12 to 18 carbon atoms long. How hard (solid, insoluble) or soft (liquid, soluble) a soap is depends on the nature of the anions and cations present in the system. Analyze how each of the following factors may affect the hardness or softness of soaps:
1. The nature of the cations. For example, Na* vs Li* vs K.
2. The length of the hydrocarbon chain. For example, 12 carbons (laureate lon), 14 carbons (myristate lon), or 18 carbons (stearate lon).
Answers
Answer:
Following are the solution to the given question:
Explanation:
For question 1:
The sodium soap containing Na+ is strong whereas the softer or liquids were potassium soap.It's hard to use lithium soap.These Na+, K+, and Li+ ions act as the hydrophilic center.Calcium and Magnesium ions could be substituted by hard water with increasing hydrophilicity.For question 2:
The hydrophobicity of its carbon chain increases but one appears weaker with only an increased length.Therefore, the laureate is hard, while the stearate is soft.The tertiary structure of a protein is a complex arrangement formed as the polypeptide chain folds and twists.
a. True
b. False
Answers
Answer:
True
Explanation:
The tertiary structure of a protein is a complex arrangement formed as the polypeptide chain folds and twists.
This folding & twisting of polypeptide chain leading to its complex structure, is true about tertiary structure of protein. It occurs due to different interactions between side chains of amino acids.
The primary forces of attraction between water molecules in H2O(l) are
1.
ionic bonds
2.
hydrogen bonds
3.
molecule-ion attractions
4.
van der Waals forces
Submit Answer
Answers
Answer:
2. Hydrogen Bonds
Explanation:
Since water is a polar covalent molecule, there is a slight negative and positive end. Due to this, the oxygen end of one water molecule gravitates towards the hydrogen molecules of another water molecule. This accounts for a bunch of weird properties of water, like why ice floats. It's also what makes water the "universal solvent," and gives all life on earth the ability to even exist.
The primary forces of attraction between water molecules in H₂O (I) are hydrogen bonds.
What kind of chemical bonding is present in water molecule?Hydrogen bonding is present in water molecule due to which it exhibits an excellent property of adhesion to itself and to other substances.The hydrogen bonding is a result of electrostatic forces of attraction which are generated by the difference in charge between slightly positive hydrogen ions and slightly negative other ions.
In case of water,hydrogen bonds are formed between neighboring hydrogen and oxygen atoms of the nearby water molecules.The attraction between water molecules itself results in a formation of a bond called as a hydrogen bond.
It is a type of covalent bond which is formed between hydrogen and oxygen atoms as one oxygen atom shares its two electrons with two hydrogen atoms .The positive charge of one hydrogen atom associates with negative charge of oxygen atom.These are weak interactions which are formed between a hydrogen atom each with a partial positive charge and an oxygen atom which is more electronegative than hydrogen.
To learn more about bonding in water click here:
https://brainly.com/question/5302822
#SPJ2
Which of the following contains the least amount (number) of molecules?
Group of answer choices
5.0 g O2
5.0 g H2O
5.0 g N2
5.0 g CO2
Answers
Has to be correct
Why is one side of the moon called "the dark side of the moon"?
O A. The moon's rotation and revolution take the same time.
B. The moon's rotation time is exactly twice the time of its revolution.
Ο Ο
C. The moon's rotation time is exactly half the time of its revolution.
Answers
Which of the first four radioisotopes in the decay series has the longest half-life? Pa-234 Th-234 U-234 U-238
Answers
Answer:
can u explain a little more then i will help like what are the four
Explanation:
Answer:
d) U-238
Explanation:
edg 2021
Which statement is true?
Answers
Answer:
A I think
Explanation:
do carbon has ions charge?
Answers
Carbon has an outer shell consisting of 4 valence electrons. ... Thus, a carbon ion can have a charge of anywhere from -4 to +4, depending on if it loses or gains electrons. Although the most common oxidation states of carbon are +4 and +2, carbon is able to make ions with oxidation states of +3, +1, -1, -2, and -3.
How do new traits enter a population?
Answers
Explanation: new borns are given traits based off their parents but also new traits that are developed off of their own. Now if that population has access to resources and a lot of it. Chances are they will become bratty,selfish and things such as.
Magma forms within the mantle as a result of _______ temperature and _______ pressure.
All rocks are made of ___________
Answers
Answer:
extreme/high
low
minerals
Explanation:
When Magma forms within the mantle what is the most often result? The most often result of this is high temperatures, and low pressure.
What is the new concentration? L
M NaCl
Answers
Answer:
Explanation:
\we must convert the mass of NaCl in grams into moles. We do this by dividing by the molecular weight of NaCl (58.4 g/mole). Then, we divide the number of moles by the total solution volume to get concentration. The NaCl solution is a 0.1 M solution.
Answer:
0.125
Explanation:
boom
Help me find out which reaction is balanced
Answers
Answer:The answer is c
Explanation:
Explanation:
Since there are three C’s on the left side, there needs to be three C’s on the right.
The coefficient in front of CO2 is now 3CO2
Next, balance the H’s.
Since there are 8 H’s on the left, there needs to be 8 on the right.
Since H2O already has two H’s you just need to multiply 2 by 4 to get 8 H’s. The coefficient in front of the H2O needs to be 4.
Finally, balance the O’s.
In 3CO2, there are 6 O’s because 3 x 2 = 6.
In 4H2O, there are 4 O’s.
6 + 4 = 10
There are 10 O’s on the right, so there needs to be 10 on the left.
Since O2 already has 2 O’s, you need to multiply it by 5 because 5 x 2 = 10.
The coefficient in front of the O2 is 5.
The coefficient in front of C3H8 is 1 because it is already balanced.
The final balanced equation has the following coefficients:
1, 5, 3, 4
Can someone please help me
Answers
Plants!
No doubt the answer is plants they are one maintaining the temperature, responsible for rains too...
What is The metric unit for volume ?
Answers
Answer:
milliliters
Explanation:
Is going to be milliliters because in the metric system of measurement,the most common unit of volume are milliliters and liters
Consider an electron with charge −e−e and mass mmm orbiting in a circle around a hydrogen nucleus (a single proton) with charge +e+e. In the classical model, the electron orbits around the nucleus, being held in orbit by the electromagnetic interaction between itself and the protons in the nucleus, much like planets orbit around the sun, being held in orbit by their gravitational interaction. When the electron is in a circular orbit, it must meet the condition for circular motion: The magnitude of the net force toward the center, FcFcF_c, is equal to mv2/rmv2/r. Given these two pieces of information, deduce the velocity vvv of the electron as it orbits around the nucleus. Express your answer in terms of eee, mmm, rrr, and ϵ0ϵ0epsilon_0, the permittivity of free space.
Answers
Answer:
Explanation:
The net force on electron is electrostatic force between electron and proton in the nucleus .
Fc = [tex]\frac{1}{4\pi\epsilon} \times \frac{e\times e}{r^2}[/tex]
This provides the centripetal force for the circular path of electron around the nucleus .
Centripetal force required = [tex]\frac{m\times v^2}{r}[/tex]
So
[tex]\frac{m\times v^2}{r}=\frac{1}{4\pi\epsilon} \times \frac{e\times e}{r^2}[/tex]
[tex]v^2=\frac{e^2}{4\pi \epsilon m r}[/tex]
[tex]v=(\frac{e^2}{4\pi \epsilon m r})^{\frac{1}{2} }[/tex]
=jskidwkjicdovfojvoeeeeeeeeeeeeeeeeeeeeeeeec
Answers
Answer:
you need a theripist or maybe just your girl if this is your question.
Explantion: you are clearly very weird but very cute and ily.
Hi, what are you doing?
Answers
Answer:
i am dancing and you ok hom
What is a empirical formula
Answers
Answer:
The empirical formula of a compound is the simplest whole number ratio of atoms of each element in the compound. It is determined using data from experiments and therefore empirical.
For example, the molecular formula of glucose is C 6H 12O 6 but the empirical formula is CH 2O.
Please mark as brainliest if answer is right
Have a great day, be safe and healthy
Thank u
XD
Answer:
a chemical formula showing the simplest ratio of elements in a compound rather than the total number of atoms in the molecule CH2O is the empirical formula for glucose.
PLEASE HELP, DUE AT 12:00
Answers
Answer:
5.C6.DExplanation:
YONG ISA PO ETO HINDI KO PO ALAM TOH
is this correct im just asking because my little brother not sure for his answer
Answers
1 point
To help you with estimating, the individual answers to A, B, C should all
have zero in the one's place. Add those answers up to get the numerical
code. Input that code to move on.*
A) 56 g NH3 can be dissolved in 100 g of water at °C
B) At 50°C, 166 g of KNO, can be dissolved in g of water to make a saturated
solution
C) At 76°C, KCI and HCI have the same solubility. What mass of each could be dissolved in
80 g of water?
Your answer
This is a required question
Back
Next
Answers
Answer:ee
Explanation:
→ Search the internet to understand the reaction between (Zno) & (HNO3).
a) Describe what happens to the first spoonful of powder when he mixes it with the acid.
b) Describe what would happen if the student kept adding zinc oxide to the nitric acid
after the reaction between the acid and metal oxide was complete.
Answers
Answer:
See Explanation
Explanation:
The equation of the reaction is;
ZnO(s) + 2HNO3(aq) --------> Zn(NO3)2(aq) + H2O(l)
a) When you first add ZnO to aqueous HNO3, the powder dissolves as it mixes with the acid.
b) If the student keeps on adding more ZnO powder to the HNO3 after the reaction between the acid and metal oxide was complete, the powder will no longer dissolve in the acid. At this point, the reaction has attained equilibrium.
Which statement best describes what occurs during a chemical reaction?
Answers
Chemical reactions involve breaking chemical bonds between reactant molecules (particles) and forming new bonds between atoms in product particles (molecules). The number of atoms before and after the chemical change is the same but the number of molecules will change
Answer:
The bonds in reactants break so that elements can rearrange form new products
Explanation:
Why is medical technology good for society?
O A. It creates more jobs to do by hand,
B. It makes bilingspitals bigger.
C. It gives patients more illnesses.
D. It helps doctors treat diseases.
Answers
Plz help plz help me will mark Brainly
Answers
Answer:
Greenhouse gases absorb some of the energy and trap it in the lower atmosphere. Less heat radiates into space, and Earth is warmer. Many greenhouse gases occur naturally. Carbon dioxide, methane, water vapor, and nitrous oxide are naturally present in Earth's atmosphere. Since some of the extra energy from a warmer atmosphere radiates back down to the surface, Earth's surface temperature rises.
Hope this helped! :)
Can anyone please help?
Answers
Answer:
Earth
Explanation:
Earth is unique in the fact that we have an oxygen-rich atmosphere
Help plz:)))I’ll mark u Brainliest
Answers
Answer:
2.475 mol of O2 formed.
Explanation:
Given 1.65 moles of KClO3 as the target amount in the reactant, used the coefficient of the balanced chemical reaction involved to determine the number of moles of O2 molecules formed.
x mole of O2 = 1.65 mol KClO3 x [(3 mol O2)/ (2 mol KClO3)] = 2.475 mol of O2
x mole of O2 formed = 2.475 mol of O2
Ksp for potassium chloride, KCl, is 21.7. A 5.0-liter container filled with otherwise-pure water, at standard temperature and pressure, contains 21.5 moles of dissolved KCl. (A) Write a chemical reaction describing the dissolution of KCl. (B) Write an equation for Ksp in terms of the reactants and products from part (A). (C) Find the solubility of KCl, in moles/liter. (D) Is the above solution undersaturated, supersaturated, or at equilibrium with respect to KCl
Answers
Answer:
See explanation below
Explanation:
a) First, as the KCl is a salt, this will ve completely dissociated by water in solution and the reaction taking place would be:
KCl <-----------> K⁺ + Cl⁻
b) According to the above reaction, the Ksp or constant of solubility product will be just the concentration of the products. This is because solid compounds do not contribute in the equilibrium expression, cause they have a constant concentration near to 1. Same thing happens with liquid compounds. So, based on this the Ksp expression for the above reaction is:
KCl(s) <-----------> K⁺(aq) + Cl⁻(aq)
Ksp = [K⁺] [Cl⁻]
c) To find this solubility we need to write the Ksp expression from above, use the given Ksp value and then solve for the solubility:
KCl(s) <-----------> K⁺(aq) + Cl⁻(aq)
Ksp = [K⁺] [Cl⁻]
21.7 = s * s
21.7 = s²
s = 4.66 mol/L
d) To know this, we need to see the concentration of the KCl in solution with the given data of 21.5 moles and the 5 liters of solution:
[K⁺] = [Cl⁻] = 21.5 / 5 = 4.3 mol/L
If we want to calculate the Ksp at this conditions we have:
Ksp2 = (4.3)²
Ksp2 = 18.49
As this value is lower than the actual Ksp value, we can conclude that this solution is undersaturated.
Hope this helps