Answers
Answer:
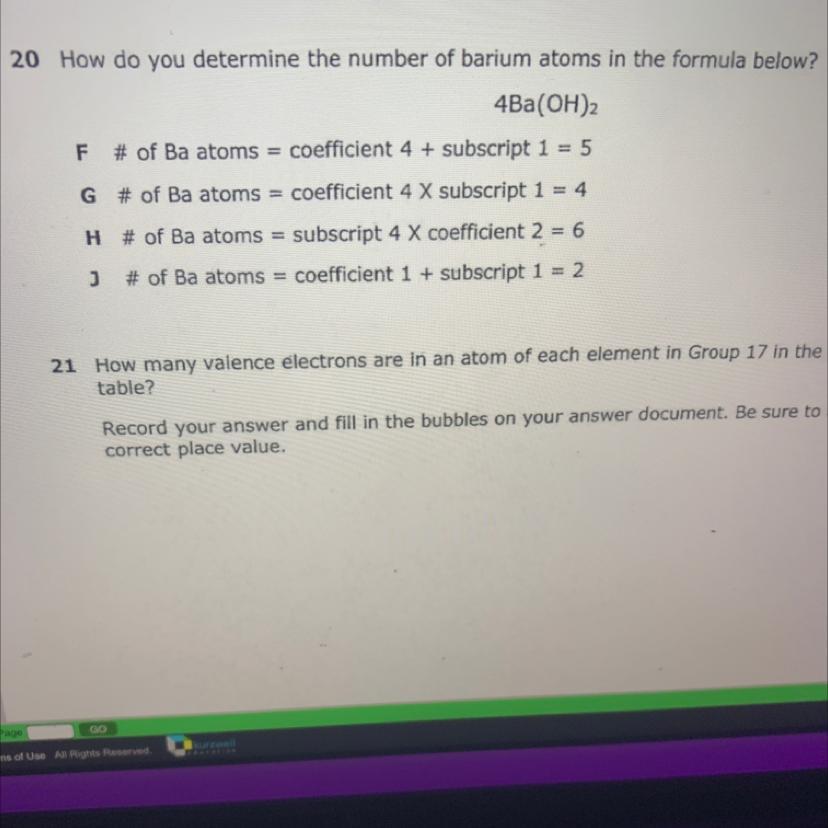
G is correct option:
# of Ba atoms = coefficient 4 × subscript 1= 4
Explanation:
Formula:
4Ba(OH)₂
G is correct option:
# of Ba atoms = coefficient 4 × subscript 1= 4
Because there are only 4 atoms of Ba in given formula.
Ba(OH)₂ contain one atom of Ba while in question there are 4 moles of Ba(OH)₂ present thus total 4×1 = 4 atoms of Ba present.
Other options are incorrect. Because,
F:
# of Ba atoms = coefficient 4 + subscript 1 = 5
This shows given formula contain 5 Ba atoms. So it is incorrect.
H:
# of Ba atoms = subscript 4 × coefficient 2 = 6
This shows that subscript is 4 which is incorrect because coefficient is 4 and subscript is 1.
j:
# of Ba atoms = subscript 1 + coefficient 1 = 2
This option shows that subscript is one which is correct but coefficient is incorrect. The coefficient of Ba is 4 and coefficient is always multiply with subscript not added. So this option is also incorrect.
Related Questions
If 25.6 mL isopropyl alcohol fully decomposes, what mass of H2 is formed? The density of isopropyl alcohol is 0.785 g/mL. g
Answers
Answer:
The correct answer is 0.67 g H₂
Explanation:
Isopropyl alcohol (C₃H₇OH) can decompose to give acetone (C₂H₆OH) and hydrogen gas (H₂) according to the following chemical equation:
C₃H₇OH (g) ⇒ C₂H₆CO(g) + H₂(g)
We can calculate the initial mass of isopropyl alcohol from the density and volume data:
density = m/V = 0.785 g/mL
⇒ m = density x V = 0.785 g/mL x 25.6 mL = 20.096 g C₃H₇OH
According to the chemical equation 1 mol of C₃H₇OH gives 1 mol H₂. The molar mass of C₃H₇OH is:
molar mass C₃H₇OH = (12 g/mol x 3) + (1 g/mol x 7) + 16 g/mol + 1 g/mol = 60 g/mol
molar mass H₂ = 1 g/mol x 2 = 2 g/mol
So, we obtain: 2 g H₂ from 60 g C₃H₇OH. We multiply this stoichiometric ratio (2 g H₂/60 g C₃H₇OH) by the initial mass of C₃H₇OH to obtain the mass of H₂ is formed:
20.096 g C₃H₇OH x (2 g H₂/60 g C₃H₇OH) = 0.6698 g ≅ 0.67 g H₂
A sample of an unknown gas weighs 0.419 grams and produced 5.00L of gas at 1.00atm (unknown gas only) and 298.15K, what is the molar mass (g/mole) of this unknown gas
Answers
Answer:
molar mass of unknown gas = 1.987 g/mol
Explanation:
First, the number of moles of the unknown gas is found
Using the ideal gas equation: PV = nRT
P = 1.00 atm, V = 5.00 L, T = 298.15 K, R = 0.082 L.atm.mol⁻¹K⁻¹
n = PV/RT
n = (1.00 atm * 5.00 L)/(298.15 K *0.082 L.atm.mol⁻¹K⁻¹)
n = 0.2109 moles
Molar mass = mass/ number of moles
molar mass = 0.419 g/ 0.2109 mols
molar mass of unknown gas = 1.987 g/mol
The molar mass of unknown gas by using ideal gas equation = 1.987 g/mol.
Ideal gas equationThis equation gives the relation between pressure, volume, temperature as given below:
[tex]PV = nRT[/tex]
P = 1.00 atm, V = 5.00 L, T = 298.15 K, R = 0.082 L.atm.mol⁻¹K⁻¹
Substitute the above values in the above equation as follows:
n = (1.00 atm * 5.00 L)/(298.15 K *0.082 L.atm.mol⁻¹K⁻¹)
n = 0.2109 moles
Formula for molar mass[tex]Molar mass = mass/ number of moles[/tex]
Calculate molar mass by using the above equation,
molar mass = 0.419 g/ 0.2109 moles
The molar mass of unknown gas = 1.987 g/mol
Find more information about ideal gas equation here,
brainly.com/question/4147359
A sample of propane, C3H8, contains 13.8 moles of carbon atoms. How many total moles of atoms does the sample contain
Answers
Answer:
[tex]Total = 50.6\ moles[/tex]
Explanation:
Given
[tex]Propane = C_3H_8[/tex]
Represent Carbon with C and Hydrogen with H
[tex]C = 13.8[/tex]
Required
Determine the total moles
First, we need to represent propane as a ratio
[tex]C_3H_8[/tex] implies
[tex]C:H = 3:8[/tex]
So, we're to first solve for H when [tex]C = 13.8[/tex]
Substitute 13.8 for C
[tex]13.8 : H = 3 : 8[/tex]
Convert to fraction
[tex]\frac{13.8}{H} = \frac{3}{8}[/tex]
Cross Multiply
[tex]3 * H = 13.8 * 8[/tex]
[tex]3 H = 110.4[/tex]
Solve for H
[tex]H = 110.4/3[/tex]
[tex]H = 36.8[/tex]
So, when
[tex]C = 13.8[/tex]
[tex]H = 36.8[/tex]
[tex]Total = C + H[/tex]
[tex]Total = 13.8 + 36.8[/tex]
[tex]Total = 50.6\ moles[/tex]
for the following reaction, provide the missing information
Answers
Answer:
19. Option B. ⁰₋₁B
20. Option D. ²¹⁰₈₄Po
Explanation:
19. ²²⁸₈₈Ra —> ²²⁸₈₉Ac + ʸₓZ
Thus, we can determine ʸₓZ as follow:
228 = 228 + y
Collect like terms
228 – 228 = y
y = 0
88 = 89 + x
Collect like terms
88 – 89 = x
x = –1
Thus,
ʸ ₓZ => ⁰₋₁Z => ⁰₋₁B
²²⁸₈₈Ra —> ²²⁸₈₉Ac + ʸₓZ
²²⁸₈₈Ra —> ²²⁸₈₉Ac + ⁰₋₁B
20. ᵘᵥX —> ²⁰⁶₈₂Pb + ⁴₂He
Thus, we can determine ᵘᵥX as follow:
u = 206 + 4
u = 210
v = 82 + 2
v = 84
Thus,
ᵘᵥX => ²¹⁰₈₄X => ²¹⁰₈₄Po
ᵘᵥX —> ²⁰⁶₈₂Pb + ⁴₂He
²¹⁰₈₄Po —> ²⁰⁶₈₂Pb + ⁴₂He
How do the test variables (independent variables) and outcome variables (dependent variables) in an experiment compare? A. The test variables (independent variables) and outcome variables (dependent variables) are the same things. B. The test variable (independent variable) controls the outcome variable (dependent variable). C. The test variable (independent variable) and outcome variable (dependent variable) have no affect on each other. D. The outcome variable (dependent variable) controls the test variable (independent variable).
Answers
Answer:
I'm on the exact same queston
Answer:
The test variable (independent variable) controls the outcome variable (dependent variable)
Explanation:
its right on study island
calculate the mass of N2 gas which has a volume 0.227 at STP
Answers
The smallest form of matter that still retains the properties of an element
Answers
Answer:
atom
Explanation:
the atom is the smallest form.
Scientists are experimenting with pure samples of isotope X which is radioactive. The sample has a mass of 20. Grams. The half-life was measured to be 232 seconds. There is a second sample that weighs 80 grams. What is the half-life of the second sample
Answers
Answer:
Explanation:
Half life of radioactive materials do not depend upon the mass of the material . It only depends upon the nature of radioactive materials . The half life of 20 g is 232 seconds . That means 20 gram will be reduced to 10 gram in 232 seconds .
Half life of 80 gram is also 232 seconds . So , 80 gram will be reduced to 40 gram in 232 second .
How to separate given mixture?
Answers
Answer:
Chromatography involves solvent separation on a solid medium.
Distillation takes advantage of differences in boiling points.
Evaporation removes a liquid from a solution to leave a solid material.
Filtration separates solids of different sizes.
Explanation:
What types of materials are better at absorbing energy from radiation?
Answers
Answer:
Explanation:
Materials are clothes and heavy rope
How many moles of precipitate will be formed when 100.0 mL of 0.200 M NaBr is reacted with excess Pb(NO₃)₂ in the following chemical reaction?
2 NaBr (aq) + Pb(NO₃)₂ (aq) → PbBr₂ (s) + 2 NaNO₃ (aq)
Answers
Answer : The number of moles of precipitate, [tex]PbBr_2[/tex] formed will be 0.01 moles.
Explanation : Given,
Concentration of NaBr = 0.200 M
Volume of solution = 100.0 mL = 0.1 L (1 L = 1000 mL)
First we have to calculate the moles of NaBr.
[tex]\text{Moles of NaBr}=\text{Concentration of NaBr}\times \text{Volume of solution in L}[/tex]
[tex]\text{Moles of NaBr}=0.200M\times 0.1L=0.02mol[/tex]
Now we have to calculate the moles of precipitate, [tex]PbBr_2[/tex] formed.
The balanced chemical reaction is:
[tex]2NaBr(aq)+Pb(NO_3)_2(aq)\rightarrow PbBr_2(s)+2NaNO_3(aq)[/tex]
From the balanced chemical reaction we conclude that:
As, 2 moles of NaBr react to give 1 mole of [tex]PbBr_2[/tex]
So, 0.02 moles of NaBr react to give [tex]\frac{0.02}{2}=0.01[/tex] mole of [tex]PbBr_2[/tex]
Therefore, the number of moles of precipitate, [tex]PbBr_2[/tex] formed will be 0.01 moles.
The number of mole of the precipitate (i.e PbBr₂) formed when 100 mL of 0.2 M NaBr react with excess Pb(NO₃)₂ is 0.01 mole
We'll begin by calculating the number of mole of NaBr in 100 mL of 0.2 M NaBr solution. This can be obtained as follow:Volume = 100 mL = 100 / 1000 = 0.1 L
Molarity of NaBr = 0.2 M
Mole of NaBr =?Mole = Molarity x Volume
Mole of NaBr = 0.2 × 0.1
Mole of NaBr = 0.02 mole Finally, we shall determine the number of mole of the precipitate (i.e PbBr₂) produced from the reaction. This can be obtained as follow:2NaBr(aq) + Pb(NO₃)₂(aq) → PbBr₂(s) + 2NaNO₃ (aq)
From the balanced equation above,
2 moles of NaBr reacted to produce 1 mole of PbBr₂.
Therefore,
0.02 mole of NaBr will react to produce = [tex]\frac{0.02}{2} \\\\[/tex] = 0.01 mole of PbBr₂.
Thus, the number of mole of the precipitate (i.e PbBr₂) produced from the reaction is 0.01 mole
Learn more: https://brainly.com/question/19572703
Question 11
4 pts
Using the formula 2H202 --> 2H2O + O2, if 7.30 moles of peroxide are
decomposed, how many moles of oxygen will be formed?
Answers
Answer:
3.65 mol O₂
Explanation:
Step 1: RxN
2H₂O₂ → 2H₂O + O₂
Step 2: Define
Given - 7.30 mol H₂O₂
Solve - x mol O₂
Step 3: Stoichiometry
[tex]7.30 \hspace{3} mol \hspace{3} H_2O_2(\frac{1 \hspace{3} mol \hspace{3} O_2}{2 \hspace{3} mol \hspace{3} H_2O_2} )[/tex] = 3.65 mol O₂
One way to represent a substance is with a chemical formula. In the formula CO2, what do the symbols Cand o refer to?
Answers
Answer:
C is for carbon and O is for oxygen
help me please loves
Answers
Answer:
K and ba
Explanation:
Answer: K and Ba
Explanation:
Which of these is the best definition of biotechnology??
A) The study of chemical processes in living things
B) the use and application of living things and science
C) the application of engineering principles to solve medical problems
D) the study of forces
Answers
Bio means life and technology is scientific knowledge for different purposes.
Sorry if this is wrong
True or False: Particles that are moving faster have a higher temperature
Answers
Answer:
true
Explanation:
I'm not sure why cause I dont know how to explain but it's TRUE
Answer:
True
Explanation:
The particles moving faster in a substance the hotter it gets.
SOMEONE PLEASE HELPPP
Answers
Which of the following evidence supports the theory of plate tectonics
Answers
Answer:
seafloor spreading
Explanation:
i took the test
Is air a homogeneous heterogeneous mixture or substance
Answers
Answer:
look at the explanation
Explanation:
The air that you breathe is a homogeneous mixture of oxygen, nitrogen, argon, and carbon dioxide, along with other elements in smaller amounts. Because each layer of the Earth's atmosphere has a different density, each layer of air is its own homogeneous mixture.
Hope this helps. :)
The empirical formula of CBr2 has a molar mass of 515.46 g/mol. What is the molecular formula of this
compound
Answers
Answer:
C3Br6
Explanation:
C= (1 X 12.011) = 12.011
Br= (2 X 79.904)= 159.808
159.808+12.011 = 171.819
515.46 divided by 171.819 = 3.00
so you mulitpy CBr2 by 3 which gives you C3Br6
Which option BEST explains how thermal equilibrium interacts with heat transfer between particles?
a
Thermal equilibrium stops the transfer of energy in just one direction when both objects reach the same temperature, but allows their particles to continue transferring that energy back and forth.
b
Thermal equilibrium always transfers energy from the hotter object to the colder one, and increases the energy and speed of moving particles in both objects as the temperature decreases.
c
Thermal equilibrium helps the transfer of energy between the particles of some materials better than others, but always stops the transfer of energy in materials like plastic and wood.
d
Thermal equilibrium quickly transfers energy back to the particles of the object that was originally hotter, and requires that the particles in both objects have reached equal energy and density.
Answers
Answer:
Thermal equilibrium stops the transfer of energy in just one direction when both objects reach the same temperature, but allows their particles to continue transferring that energy back and forth.
Explanation:
a. The transfer of energy occurs until both objects reach thermal equilibrium. But particles are always moving and crashing with each other. TRUE.
b. The heat transfer occurs from the hotter object to the colder one but moving of particles descreases with temperature decreasing. FALSE.
c. Plastic and wood have a poor quality to transfer energy but there is no material that "stops" perfectly the transfer of energy. FALSE.
d. The heat is transferred from the particles of the hotter object to the particles of the colder one. FALSE
When does carbon dioxide absorb the most heat energy?
during freezing
during deposition
during sublimation
during condensation
Answers
This has been posted on here before so you could’ve searched it lol.
Best of luck :))
Answer:
during sublimation
Explanation:
just took the test
What can the chemical formula tell us about a compound?
Answers
Answer:
A chemical formula tells us the number of atoms of each element that is in a compound. It contains the symbols of the atoms for the elements present in the compound as well as how many there are for each element in the form of subscripts.
Hope this helps! please mark me brainliest!
God bless :)
The smallest unit of an element that can exist either alone or in combination with other such particles of the same or different elements is the
А.
electron
B
proton
neutron
D
atom
Answers
Answer:
The answer is D - Atom
The smallest unit of an element that can exist either alone or in combination with other such particles of the same or different elements is the atom.
What is an atom?
An atom is defined as the smallest unit of matter which forms an element. Every form of matter whether solid,liquid , gas consists of atoms . Each atom has a nucleus which is composed of protons and neutrons and shells in which the electrons revolve.
The protons are positively charged and neutrons are neutral and hence the nucleus is positively charged. The electrons which revolve around the nucleus are negatively charged and hence the atom as a whole is neutral and stable due to presence of oppositely charged particles.
Atoms of the same element are similar as they have number of sub- atomic particles which on combination do not alter the chemical properties of the substances.
Learn more about atom,here:
https://brainly.com/question/13654549
#SPJ2
Which of the following is a good definition of matter?
O A. Anything that is made up of light and gravity
O B. Anything that has mass and takes up space
O C. Anything that produces heat and mass
O D. Anything that has energy and creates heat
Answers
Answer:
B
Explanation:
I did the question before and got it right.
B
Explanation:
I got it right on my test.
What is the Kc equilibrium-constant expression for the following equilibrium? S8(s) + 24F2(g) 8SF6(g)
Answers
Answer:
[tex]Kc=\frac{[SF_6]^8}{[F_2]^2^4}[/tex]
Explanation:
Hello.
In this case, for the undergoing chemical reaction:
[tex]S_8(s) + 24F_2(g) \rightleftharpoons 8SF_6(g)[/tex]
We consider the law of mass action in order to write the equilibrium expression yet we do not include S8 as it is solid and make sure we power each gaseous species to its corresponding stoichiometric coeffient (24 for F2 and 8 for SF6), thus we obtain:
[tex]Kc=\frac{[SF_6]^8}{[F_2]^2^4}[/tex]
Best regards!
Which of the following is NOT a strong electrolyte?
:
Answers:
Na2SO4
KI
CaCl2
LiOH
C6H1206
Answers
Answer:
C6H1206
Explanation:
C6H12O6 is a monomer of carbohydrates also known as glucose, so it is not an electrolyte at all.
Which of the following is an Elementary compound?
A. CO2
B. N2
C. SO2
D. H2S
heeeeeeeeeelp please please please
Answers
Answer:
Explanation:
In my opinion the answer should be SO2
Answer:
a should be answer i think.
A chemist decomposes samples of several compounds; the masses of their constituent elements are listed. Calculate the empirical formula for each compound.
a. 1.245 g Ni, 5.381 g I,
b. 2.677 g Ba, 3.115 g Br,
c. 2.128 g Be, 7.557 g S, 15.107 g
Answers
Answer:
you can see the empirical formula at the pic
The empirical formula for compound (a) is NiI2, (b) is BaBr2 and (c) is BeS.
What is empirical formula?
Empirical formula of a compound is defined as the simplest whole number ratio of atoms present in a compound.
(a) 1.245 g Ni : 5.381 g I
Mole of Ni ; Mole of I = 1.245/59 : 5.381/127 = 0.02 : 0.04 = 1:2
So the formula is NiI2
(b) 2.677 g Ba : 3.115 g Br
Mole of Ba : Mole of Br = 2.677/137 : 3.115/60 = 0.019 : 0.038
= 0.02 : 0.04 = 1:2
So the formula is BaBr2
(c) 2.128 g Be : 7.557 g S
Mole of Be : Mole of S = 2.128/9 : 7.557/32 = 0.2 : 0.2 = 1:1
So the formula is BeS
Thus, empirical formula for compound (a) is NiI2, (b) is BaBr2 and (c) is BeS.
To learn more about empirical formula, refer to the link below:
https://brainly.com/question/11588623
#SPJ2
What happens when the elements in group 2 react with water?
Answers
Answer:
The Group 2 metals become more reactive towards the water as you go down the Group.
Explanation:
These all react with cold water with increasing vigour to give the metal hydroxide and hydrogen. ... You get less precipitate as you go down the Group because more of the hydroxide dissolves in the water. Summary of the trend in reactivity.
Please mark me brainliest! hope this helped!
God bless!